 INTERNATIONAL STANDARD ISO 6579-1
INTERNATIONAL STANDARD ISO 6579-1
A list of all parts in the ISO 6579 series can be found on the ISO website. vi. © ISO 2017 – All rights reserved. iTeh STANDARD PREVIEW. (standards.iteh.ai).
 Safety Data Sheet: EN ISO 6579 ISO 10273
Safety Data Sheet: EN ISO 6579 ISO 10273
https://www.carlroth.com/medias/SDB-X928-IE-EN.pdf?context=bWFzdGVyfHNlY3VyaXR5RGF0YXNoZWV0c3wyMjcwNTJ8YXBwbGljYXRpb24vcGRmfHNlY3VyaXR5RGF0YXNoZWV0cy9oMzIvaGE2LzkwMzk4MTg2ODY0OTQucGRmfDM0MGVmODQ1ZDNjMDNiZWVkMjA3MmM1OWYzMGI4NTY1N2FjN2Y0ZDYxMTIzNWNlNDgzMzNhMjU4ZDZhYTc5ZTI
 (84-4) 37 ISO 6579-1:2017(E)
(84-4) 37 ISO 6579-1:2017(E)
For characterization of Salmonella strains full serotyping is needed. Guidance for serotyping is described in ISO/TR 6579-3[24]. For some of the confirmation
 SIST EN ISO 6579-1:2017/oprA1:2019
SIST EN ISO 6579-1:2017/oprA1:2019
1 sept. 2019 EN ISO 6579-1:2017/prA1. 07.100.30. Mikrobiologija živil. Food microbiology. ICS: SIST EN ISO 6579-1:2017/oprA1:2019 en. 2003-01.Slovenski ...
 REGULAMENTUL (UE) 2019/ 229 AL COMISIEI - din 7 februarie
REGULAMENTUL (UE) 2019/ 229 AL COMISIEI - din 7 februarie
8 feb. 2019 ... ISO 6579” se înlocuiesc cu termenii „EN ISO 6579-1”;. (iii) în rândul 2.1.5 termenii „EN/ISO 6579 (pentru detecție)” se înlocuiesc cu ...
 Validation of EN ISO 6579-1 - Microbiology of the food chain
Validation of EN ISO 6579-1 - Microbiology of the food chain
stage (EN ISO 6579:2002/Amd.1:2007) and its performance characteristics in the new part 1. 39 of EN ISO 6579. The 2002 version of EN ISO 6579 already
 ReadyTube™ 1000 Buffered Peptone Water acc. ISO 6579 ISO
ReadyTube™ 1000 Buffered Peptone Water acc. ISO 6579 ISO
This culture medium complies with the specifications given by EN ISO 6579 EN ISO/FDIS. 6579-1
 MSRV (Modified Semi‑Solid RAPPAPORT‑ VASSILIADIS) Medium
MSRV (Modified Semi‑Solid RAPPAPORT‑ VASSILIADIS) Medium
EN ISO 6579/Amd 1 describes the usage of MSRV medium for the detection of Salmonellae in samples from the primary production stage. EN ISO FDIS 6579-1 gives
 ReadyTube™ 225 Buffered Peptone Water acc. ISO 6579 ISO 6887
ReadyTube™ 225 Buffered Peptone Water acc. ISO 6579 ISO 6887
This culture medium complies with the specifications given by EN ISO 6579 EN ISO/FDIS. 6579-1
 ANALIZE MICROBIOLOGICE ACREDITATE RENAR
ANALIZE MICROBIOLOGICE ACREDITATE RENAR
SR EN ISO 6579 :2003;. SR EN ISO 6579 :2003/AC /2006 ; teste de sanitatie. PS-LML-CM-20 ed.03 rev.01. PS-LML-CM-17. SR EN ISO 6785/2007. PS-LML-CM-04 PS-LML-CM
 ?ISO 6579-1:2017?
?ISO 6579-1:2017?
6579-1. First edition. 2017-02. Reference number. ISO 6579-1:2017(E) Guidance for serotyping of Salmonella spp. is described in ISO/TR 6579-3.[24].
 standard iso 6579-1
standard iso 6579-1
6579-1. First edition. 2017-02. Reference number. ISO 6579-1:2017(E). This preview is downloaded from www.sis.se. Buy the entire standard via
 SIST EN ISO 6579-1:2017/oprA1:2019
SIST EN ISO 6579-1:2017/oprA1:2019
1 sept. 2019 statusu Dodatka D in popravek MSRV ter SC (ISO 6579-1:2017/DAM 1:2019). Microbiology of the food chain - Horizontal method for the detection ...
 Technical Data Sheet GranuCult™ Buffered Peptone Water acc. ISO
Technical Data Sheet GranuCult™ Buffered Peptone Water acc. ISO
ISO 6579 ISO 21528
 Validation of EN ISO 6579-1 - Microbiology of the food chain
Validation of EN ISO 6579-1 - Microbiology of the food chain
Validation of EN ISO 6579-1 - Microbiology of the food chain - Horizontal method for. 3 the detection enumeration and serotyping of Salmonella - Part 1
 Detection of Salmonella species National Infection Service Food
Detection of Salmonella species National Infection Service Food
The method described here is based on BS EN ISO 6579-1:2017 and includes the method for detection of Salmonella Typhi and S.Parapyphi as detailed in Annex D
 SUMMARY OF VALIDATION UNE-EN ISO 16140 METHOD EASY
SUMMARY OF VALIDATION UNE-EN ISO 16140 METHOD EASY
Reference Methods: UNE-EN ISO 6579: 2003. Microbiology of Food and animal feeding sutffs. Horizontal method for the detection of Salmonella spp.
 Technical Data Sheet GranuCult™ RVS (RAPPAPORT
Technical Data Sheet GranuCult™ RVS (RAPPAPORT
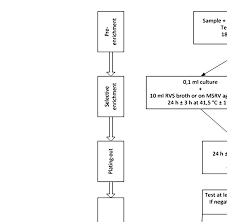
According to EN ISO 6579 transfer 0.1 ml of the culture obtained in the pre- enrichment (Buffered Peptone Water) to a tube containing 10 ml of RVS broth.
 GranuCult® prime Hektoen enteric (HE) agar acc. ISO 21567 FDA
GranuCult® prime Hektoen enteric (HE) agar acc. ISO 21567 FDA
30 nov. 2020 This culture medium complies with the specifications given by EN ISO 21567 FDA-BAM Medium. M61
 Readybag® Buffered Peptone Water acc. ISO 6579 ISO 21528
Readybag® Buffered Peptone Water acc. ISO 6579 ISO 21528
https://www.merckmillipore.com/INTERSHOP/web/WFS/Merck-JP-Site/ja_JP/-/JPY/ShowDocument-File?ProductSKU=MDA_CHEM-100901&DocumentId=201512.273.ProNet&DocumentUID=46316172&DocumentType=DS&Language=EN&Country=NF&Origin=PDP
 NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Store
NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Store
>NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Storehttps://cdn standards iteh ai/ /ISO-6579-1-2017 pdf · Fichier PDF
 INTERNATIONAL ISO STANDARD 6579-1 - iTeh Standards Store
INTERNATIONAL ISO STANDARD 6579-1 - iTeh Standards Store
>INTERNATIONAL ISO STANDARD 6579-1 - iTeh Standards Storehttps://cdn standards iteh ai/ /ISO-6579-1-2017-Amd-1-2020 pdf · Fichier PDF
 INTERNATIONAL ISO STANDARD 6579-1
INTERNATIONAL ISO STANDARD 6579-1
>INTERNATIONAL ISO STANDARD 6579-1
 INTERNATIONAL ISO STANDARD 6579-1
INTERNATIONAL ISO STANDARD 6579-1
>INTERNATIONAL ISO STANDARD 6579-1https://cdn standards iteh ai/ /ISO-6579-1-2017 pdf · Fichier PDF
 NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Store
NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Store
>NORME ISO INTERNATIONALE 6579-1 - iTeh Standards Storehttps://cdn standards iteh ai/ /ISO-6579-1-2017-Amd-1-2020 pdf · Fichier PDF
What is EN ISO 6579:2002?
EN ISO 6579:2002 - Microbiology of food and animal feeding stuffs - Horizontal method for the detection of Salmonella spp (ISO 6579:2002)
What is ISO ISO 6579-1?
ISO 6579-1:2017(E) Figure A.1 — Diagram of procedure for detection of Salmonella in food, animal feed, and environmental samples from the food production area
Where can I find a list of ISO 6579 parts?
A list of all parts in the ISO 6579 series can be found on the ISO website. This document describes a horizontal method for the detection of Salmonella spp. in food (including milk and milk products, originally described in ISO 6785), in animal feed, in animal faeces, and in
Miguel Yuste, 12 - 4ª planta
28037 Madrid
(+34) 914 401 224 aenorlaboratorio@aenor.com www.aenor.comSUMMARY OF VALIDATION UNE-EN ISO 16140
METHOD EASY QFAST®
DETERMINATION OF SALMONELLA spp.
CUALITATIVE METHOD
Edition 4 (08-05-2017)
Page 2 of 45
INDEX1.- INTRODUCTION 3
2.- ALTERNATIVE METHOD AND REFERENCE METHOD 4
2.1.- PRINCIPLE OF THE ALTERNATIVE METHOD EASY QFAST
SALMONELLA
42.2.- PRINCIPLE AND PROTOCOL OF ALTERNATIVE METHOD HARD EASY
VETERINARY QFAST 8
2.3.- REFERENCE METHOD
133.- COMPARATIVE STUDY OF THE METHODS 13
3.1.-RELATIVE ACCURACY, RELATIVE SPECIFICITY AND RELATIVE
SENSITIVITY 13
3.2.- RELATIVE DETECTION LIMIT (LDR) 27
3.3.- INCLUSIVITY AND EXCLUSIVITY 28
4.- COLABORATORY STUDY 30
4.1.- ORGANIZATION OF THE STUDY 30
4.2.- RESULTS OF THE ANALYSIS 32
4.3.- CALCULATIONS 33
5.- INTERPRETATION 37
6.- AUDITS 40
7.- CONCLUSION
41ANNEXES
Annexe 1 ± Protocol of alternative method EASY QFast® 42Annexe 2 ± Reference method ISO 6579:2002-Microbiology of Food and animal feeding stuffs. 44 Horizontal method for the detection of Salmonella spp. Annexe 3 ± Reference method ISO 6579/A1-Microbiology of Food and animal feeding stuffs.
45 Horizontal method for the detection of Salmonella spp. Amedement
1: Annex D.
Detection of Salmonella spp in faeces ans enviromental samples from primary production stagePage 3 of 45
1.- INTRODUCTION
The results obtained that appear in the corresponding tables as well as their treatment have been
performed according to the norm UNE-EN ISO 16140. Manufacturer: iMICROQ, Integrated Microsystems for Quality of Life, S.L.Polígon Industrial Riu Clar
C/ Ferro 6 (nau 7)
43006 Tarragona, Spain
Certification Body: Asociación Española de Normalización y Certificación (AENOR)C/ Génova 6
28004 Madrid, España
Expert laboratory: AENORlaboratorio
Miguel Yuste, 12, 4ª planta.
28037 Madrid
Method to validate: Method Easy QFast®, Fast method for detection of Salmonella spp., fully validated
according UNE-EN ISO 16140:2003. ³0LŃURNLRORgy of Food and animal feeding stuffs. Protocol for validation
of alternative methods´ 81(-EN ISO 16140:2003/A1:2012).The validation has comprised two stages:
Comparative study of the alternative method versus the reference method performed byAENORLaboratory
Collaborative study of each of the two methods: Alternative method and Reference method.Validation Reference: UNE-EN ISO 16140:2003. Microbiology of Food and animal feeding stuffs. Protocol
for validation of alternative methods.UNE-EN ISO 16140:2003/A1:2012
Reference Methods: UNE-EN ISO 6579: 2003. Microbiology of Food and animal feeding sutffs. Horizontal method for the detection of Salmonella spp. UNE-EN ISO 6579 (2003). Erratum: 2007 V2. Horizontal method for the detection of Salmonella spp. UNE-EN ISO 6579:2003/A1:2007. Microbiology of Food and animal feeding sutffs. Horizontal method forthe detection of Salmonella spp. Amendement 1: Annex D. Detection of Salmonella spp. in animal faeces
and in environmental samples from the primary production stage.Scope of the validation:
Animal feed samples.
Veterinary samples (faeces, shoe covers,necks)
Environmental samples originating from primary production (poultry industry). General Food: fruits and vegetables, dairy products and various products, including spices, mayonnaise and eggs; and meat.Page 4 of 45
2.- ALTERNATIVE METHOD AND REFERENCE METHOD
2.1.- PRINCIPLE AND PROTOCOL OF ALTERNATIVE METHOD EASY QFAST SALMONELLA EXCEPT
FOR VETERINARY SAMPLES
2.1.1.- PRINCIPLE
a) Pre-enrichment in medium 1 Add to 225 ml of pre-enrichment medium the adequate quantity of sample and incubate at37ºC ± 1ºC for 16 ±1 hours.
b) Enrichment in medium 2:Take 10 µl of the pre-enriched sample and incubate at 41.5 ºC ± 1ºC for 7 hours ± 1 hour.
c) Reading Add to the eppendorf tube, 1 drop of reaction solution and 1 drop of the enrichment mediumwith the sample with a steril Pasteur pipete; mix and vortex and incubate at 37 ºC ± 1ºC for 30
to 60 minutes. Add 10 µl in the sensor electromagnetic reader.2.1.2.-PROTOCOL
a) Pre-enrichment1- Preparation of the samples, in the laminar flow cabinet or when it is not available, a Bunsen
Burner.
Animal Feeding Samples
Weigh, with a precision of ± 1%, 25 g of sample in the flask that contains Soft medium 1. If the Soft media does not contain supplements, add them.Environmental Samples (supports, Box covers)
Supports:
Submerge the 4 shoe covers in the flask that contains Hard medium 1. If the Hard medium does not contain supplements, add them. Box covers:
Cut the sample to obtain 25 g of sample, with sterile material, submerge in the flask that contains Hard medium 1. If the Hard medium does not contain supplements, add them.Food Samples
Weigh, with a precision of ± 1%, 25 g of sample in the flask that contains Soft medium 1. If the Soft medium does not contain supplements, add them. In the case of meat products use the Hard medium.Page 5 of 45
In the case of foods that contain inhibitor substances, such as spices, the preparation of the
samples is carried out according to the norm UNE-EN ISO 6887-4 2003 ³0LŃURNLRORJ\ RI IRRG IRU human use and animal food. Preparation of the samples for analysis, initial suspension and decimal dilutions for microbiological examination. Section 4: Specific rules for the preparation of products different to milk and lactose product, meat and meat products and fish and fished SURGXŃPV´B2- Once the samples are prepared as indicated in point 1, proceed in the same manner for all of
them.Agitate manually and horizontally the flask that contains the sample and the Pre-enrichment
medium. Incubate the sample at 37ºC ± 1 ºC for 16 hours ± 1 hour. b) Enrichment in medium 2 Wait until the enrichment medium reaches room temperature (between 18 and 25ºC) before use. Remove the sample from the incubator and agitate the flask manually and horizontally. Then, take previously 1 ml from the flask, with a pipette Pasteur and add to a sterile Eppendorftube; subsequently agitate and vortex. Take 10 µl of this tube, with fixed pipette and add to
medium 2.Agitate with the vortex. Incubate at 41.5 ºC ± 1ºC for 7 hours ± 1 hour, in a shaking incubator
iMICROQ. c) Reading Reaction Wait until the solution reached room temperature (between 18ºC and 25ºC) before use. Add to an Eppendorf tube a drop of the reaction solution and a drop of enrichment medium withthe sample using a Pipette Pasteur, agitate with the vortex and incubate at 37ºC ± 1 ºC for 30-60
minutes in the shaking incubator iMICROQ. In the case where the reaction solution is concetrated, a drop of the reaction solution is added to the eppendorf contained in medium 2. Remove the eppendorf from the skaking incubator and take 10 µl with a fixed micropippette and add to the sensor, that has been previously cooled down, that has previously been fixed in the electronic reader. Read by pressing the start button. In the case that the results of the reader indicates Positive, take with inoculating loop, from the same eppendorf tube and inoculate on a plate with two selective agar: XLD and ASAP.Page 6 of 45
d) Expression of the results The PRESENCE of Salmonella spp. is considered in the part of the analysis (specifying the weight in grams, of the sample analysed or surface of the sample), when:The electrochemical sensor indicates: Positive
1RPH LI POH VHQVRU LQGLŃMPHV ³UHPHVP´ PHMVXUH MJMLQ RLPO MQRPOHU VHQVRU RLPO POH UHPMLQLQJ VMPSOH
in the eppendorf, taking the second measurement as the definitive one. A result with a positive value indicates that the sample is contaminated with Salmonella.In this case, the result should be confirmed.
The ABSENCE of Salmonella spp. in the part analysed (specifying the weight in grams, of the
sample or the surface sampled), when:The electrochemical reader indicates: Negative
A result with a Negative test indicates that the sample does not contain Salmonella at a
concentration lower than the detection limitConfirmation of positive results
All of the positive results of the method QFast® Salmonella should be confirmed. The confirmation should be carried out using the liquid reaction prior to reading and should be started after the read. Isolate specific plates, as for example XLD and ASAP. Incubate the plates at37ºC for 24 hours. Confirm the suspicious colonies via adequate biochemical and/or serological
tests.Confirmation of positive results
All of the positive results of the method Easy QFast® Salmonella should be confirmed. The confirmation should be carried out using the liquid reaction prior to reading and should be started after the read. Isolate specific plates, as for example XLD and ASAP. Incubate the plates at37ºC for 24 hours. Confirm the suspicious colonies via adequate biochemical and/or serological
tests.Page 7 of 45
Conservation of the enrichment medium and the reagents of the Easy QFast® methodMedium 1: Pre-enrichment medium (HARD):
Presentation in flasks with 225 ml of medium con supplements. Preservation between 2 and 8 ºC.
Expiration date 15 months from production.
Presentation in flasks with 225 ml of medium without supplements. Preservation between 2 and 8ºC. Expiration date 15 months from production.
Presentation bottles of dehydrated pre-enrichment medium without supplements. Preservation between 4 and 30 ºC. Expiration date 3 years from production. Medium Supplement 1 in glass packaging with a dropper. Preservation between 4 and 30 ºC.
Expiration date 15 months from production.
Medium 1: Pre-enrichment medium (SOFT):
Presentation in flasks with 225 ml of the medium with supplements. Preservation between 2 and 8ºC. Expiration date 15 months from production.
Presentation in bottles of dehydrated pre-enrichment medium without supplements. Preservation between 4 and 30 ºC. Expiration date 3 years from production. Supplement medium 1 in glass packaging with a dropper (only for the dehydrated format).
Conservation between 4 and 30 ºC. Expiration date 1 year after production.Medium 2: Enrichment medium
Presentation in eppendorf tubes with 1 ml of medium. Preservation between 2 and 8 ºC. Expiration
date 15 months from production. Dehydrated format in aluminium bags of 2.8 g. Preservation between 4 and 30 ºC. Expiration date
three years from production.Reaction Solution:
Presentation a bottle of 5 ml with a dropper. Preservation between -21 y -5ºC. Expiration date one
year from production. Presentation in two bottles of 3 ml with a dropper; one with the dehydrated reaction solution and
another with the diluent solution. Preservation between -21 and -5ºC. Expiration date one year from production.Sensor:
Presentation in plastic packaging with silicagel. Preservation between 2 and 8 ºC. Expiration date
one year from production.Page 8 of 45
2.2.- PRINCIPLE AND PROTOCOL OF ALTERNATIVE METHOD HARD EASY VETERINARY QFast
SALMONELLA FOR VETERINARY SAMPLES .
2.2.1.- PRINCIPLE
a) Pre-enrichment in Medium 1 and Medium 1-P This step is necessary for proper recovery and pre-enrichment of Salmonella, while limiting the excessive proliferation of other accompanying flora. Pre-enrichment is done in two stages. b) Enrichment in Medium 2 This second incubation step reduces significantly the accompanying flora, maintaining or increasing the population of Salmonella. c) Reading Using specific and typical enzymes of Salmonella isolated in the preceding step and an enzymatic substrate, a measurable and quantifiable signal is obtained on the electronic reader to detect the presence or absence of Salmonella in the sample. Positive results must be confirmed at a later stage.2.2.2.-PROTOCOL
a) First pre-enrichment stage in liquid medium (MEDIUM 1 HARD) Wait for the pre-enrichment medium to reach room temperature (between 18 and 25 °C) before use. Add 1ml of SUPPLEMENT FOR MEDIUM 1 HARD in the container of MEDIUM 1 HARD.Page 9 of 45
Veterinary Samples (faeces, neck, shoe covers)
Faeces:
Weigh, with a precision of ± 1%, 25 g of sample in the flask that contains Hard medium 1
previously supplemented. Neck:
Weigh, with a precision of ± 1%, 25 g of sample in the flask that contains Hard medium 1
previously supplemented. Shoe covers:
Add the number of the shoe covers necessary in the flask that contains Hard medium 1 previously supplemented.Mix manually the plastic container that contains the sample and the pre-enrichment medium,
strongly and horizontally while maintaining the cap up and suitably closed. Incubate the sample at 37ºC ± 1 ºC for 18 hours ± 1 hour. NOTE: If required, the sample incubated in this point a) can be refrigerated (between 2 and 8ºC) for a maximum of 48 hours before continuing with the procedure. b) Second pre-enrichment stage in liquid medium (MEDIUM 1-P) NOTE: The first time you use the kit or before performing the test, dispense aseptically 1 ml of MEDIUM 1-P into the sterile microtubes supplied, and keep them refrigerated if they will not be used immediately. Dispensed medium can be stored refrigerated (2 °C to 8 °C) and away from light. Wait for the microtube with MEDIUM 1-P previously dispensed to reach room temperature (between 18 and 25 °C) before use. Remove the container containing the sample and the pre-enrichment medium MEDIUM 1 HARD from the incubator and mix manually and horizontally.7MNH 100 NjO RI POe previous container with a micropipette and transferred to the microtube that
contains MEDIUM 1-P. The solution is inverted and incubated at 41,5ºC ± 1ºC for 7 hours ± 1
hour, in iMICROQ incubator under agitation. NOTE: In the event that due to the nature of the sample, for the presence of foam or particulatematter or difficulty taking 100 µl with a micropipette directly from the container obtained in second
paragraph of the point b), take under sterile conditions 1 ml from the container obtained in this point, place it in a sterile container and stir with a vortex. Then follow the instructions in third paragraph of the point b).Page 10 of 45
c) Enrichment in selective liquid medium (MEDIUM 2) NOTE: Take into account the provisions of Annex 1 before using MEDIUM 2 provided. Wait for the enrichment medium to reach room temperature (between 18 and 25 °C) before use.Add under sterile conditions 1mL of MEDIUM 2 in the sterile microtube supplied. Remove the
container containing the sample and the second pre-enrichment medium (MEDIUM 1-P) from the incubator.7MNH 10 NjO RI POH SUHYLRXV ŃRQPMLQHU RLPO M PLŃURSLSHPPH MQG PUMQVIHUUHG PR 0(GH80 2B 7OH VROXPLRQ
is inverted and incubated at 41,5ºC ± 1ºC for 18 hours ± 1 hour, in iMICROQ incubator under
agitation. d) Reaction and reading NOTE: The first time you use the kit, reconstitute the reaction solution according to the provisions of Annex 1 and the product label. Wait for the reaction solution (REACTION SOLUTION EASY) and the SENSOR to reach room temperature (between 18 and 25 °C) before use. Add in the microtube containing the sample with medium 2 after the incubation, one droplet of REACTION SOLUTION EASY, vortex and incubate at37ºC ± 1 ºC for 30 minutes in iMICROQ incubator under agitation.
Afterwards, the tube is UHPRYHG IURP POH LQŃXNMPRU MQG LPPHGLMPHO\ ORPRJHQLVHG MQG 10 NjO RI the solution is taken with a micropipette and is introduced in a SENSOR that has been placed in the electronic reader. The reading is carried out by pressing Start key in the electronic reader. e) Expression of the results The PRESENCE of Salmonella spp. is considered in the part of the analysis (specifying the weight in grams, of the sample analysed or surface of the sample), when:The electrochemical sensor indicates: Positive
Note: LI POH VHQVRU LQGLŃMPHV ³UHPHVP´ PHMVXUH MJMLQ RLPO MQRPOHU VHQVRU RLPO POH UHPMLQLQJ VMPSOH
in the eppendorf, taking the second measurement as the definitive one. A result with a positive value indicates that the sample is contaminated with Salmonella.In this case, the result should be confirmed.
The ABSENCE of Salmonella spp. in the part analysed (specifying the weight in grams, of the
sample or the surface sampled), when:The electrochemical reader indicates: Negative
Page 11 of 45
A result with a Negative test indicates that the sample does not contain Salmonella at a
concentration lower than the detection limitConfirmation of positive results
All of the positive results of the method QFast® Salmonella should be confirmed. The confirmation should be carried out using the liquid reaction prior to reading and should be started after the read. Isolate specific plates, as for example XLD and ASAP. Incubate the plates at37ºC for 24 hours. Confirm the suspicious colonies via adequate biochemical and/or serological
tests.Confirmation of positive results
All of the positive results of the method Easy QFast® Salmonella should be confirmed. The confirmation should be carried out using the liquid reaction prior to reading and should be started after the read. Isolate specific plates, as for example XLD and ASAP. Incubate the plates at37ºC for 24 hours. Confirm the suspicious colonies via adequate biochemical and/or serological
tests. Conservation of the enrichment medium and the reagents of the Hard Easy Veterinary QFast®Salmonella method
Medium 1: Pre-enrichment medium (HARD):
Presentation in flasks with 225 ml of medium con supplements. Preservation between 2 and 8 ºC.
Expiration date 15 months from production.
Presentation in flasks with 225 ml of medium without supplements. Preservation between 2 and 8ºC. Expiration date 15 months from production.
Presentation bottles of dehydrated pre-enrichment media without supplements. Preservation
between 4 and 30 ºC. Expiration date 3 years from production. Medium Supplement 1 in glass packaging with a dropper. Preservation between 4 and 30 ºC.
Expiration date 15 months from production.
Medium 1-P: Second pre-enrichment:
Liquid medium in plastic bottle (50ml). Preservation between 2 and 8 ºC. Expiration date 15
months from fabrication.Medium 2: Enrichment medium
Presentation in eppendorf tubes with 1 ml of medium. Preservation between 2 and 8 ºC. Expiration
date 15 months from production. Presentation in plastic bottle of 50 ml. Preservation between 2 and 8 ºC. Expiration date 15 months
from production.Page 12 of 45
Reaction Solution:
Presentation a bottle of 5 ml with a dropper. Preservation between -21 y -5ºC. Expiration date one
year from production. Presentation in two bottles of 3 ml with a dropper; one with the dehydrated reaction solution and
another with the diluent solution. Preservation between -21 and -5ºC. Expiration date one year from production.Sensor:
Presentation in plastic packaging with silicagel. Preservation between 2 and 8 ºC. Expiration date
one year from production.The following table presents a summary of the description of the method together with the application field
and the corresponding reference method. Table 1: Summary of the description of the method with the application fieldDescription Application Field Reference method
Método alternativo EASY
QFast® HARD para la
detección rápida deSalmonella spp
Veterinary samples from the poultry
industryEnvironmental samples from primary
production (poultry) MeatsNorm UNE-EN ISO
6579:2003/A1:2007
Norm UNE-EN ISO 6579:2003
Alternative Method HARD
EASY Veterinary QFast®
Salmonella SOFT for fast
detection of Salmonella sppVeterinary samples
Norm UNE-EN ISO
6579:2003/A1:2007
Alternative Method EASY
QFast® Salmonella SOFT
for fast detection ofquotesdbs_dbs21.pdfusesText_27[PDF] ena adresse
[PDF] ena france 2017
[PDF] ena omnivox
[PDF] ena rabat concours 2017
[PDF] ena rabat inscription 2017
[PDF] ena rabat seuil 2016
[PDF] ena rabat seuil 2017
[PDF] enafor
[PDF] encadrant ou manager
[PDF] encadrant technique pédagogique et social
[PDF] encadrement d'amplitude 1
[PDF] encadrement d'un étudiant infirmier par un autre étudiant
[PDF] encadrement d'un stagiaire aide soignant
[PDF] encadrement différencié 2016
