 BIOGEOCHEMICAL CYCLES
BIOGEOCHEMICAL CYCLES
NITROGEN FIXATION__ Process in which nitrogen gas from the atmosphere is converted into ammonia by bacteria that live in the soil and on the roots of plants
 Cycles worksheet
Cycles worksheet
Cycles worksheet. Please answer the following using the words in the text box. Carbon Cycle. 1. Plants use CO2 in the process of. to make
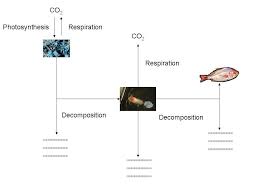 Lesson 4: The Biogeochemical Cycle
Lesson 4: The Biogeochemical Cycle
Place the carbon cycle cards included in the Lesson 4 folder (File: Carbon Cycle. Cards.pdf) within their designated spheres. 3. Assign a student to play the
 Carbon and Other Biogeochemical Cycles
Carbon and Other Biogeochemical Cycles
pdf/DynamicVegetationModels.pdf). CLM4C = Community Land Model for Carbon; CLM4CN = Community Land Model for Carbon–Nitrogen; GUESS = General Ecosystem ...
 ERTH/ENVS 3601.03 – Global Biogeochemical Cycles Winter 2019
ERTH/ENVS 3601.03 – Global Biogeochemical Cycles Winter 2019
Jan 16 2019 (available in PDF format in Dalhousie Libraries online). COMMUNICATION. - The ... Quiz #11 on Chapter 10. Chapter 13 and 14. 23 W12 Fri Apr 5.
 WATER CARBON AND NITROGEN CYCLE WORKSHEET
WATER CARBON AND NITROGEN CYCLE WORKSHEET
This plate and the ones that follow trace the pathways of several elements through biogeochemical cycles. The prime focus of this plate is on the arrows that.
 Lesson 5: The Dead Zone
Lesson 5: The Dead Zone
Ask students to recall the preceding lesson on biogeochemical cycles or give students a Try to leave enough time before the bowl quiz to go over the answers.
 SCIENCE 8 MODULE 4: LIVING THINGS AND THEIR
SCIENCE 8 MODULE 4: LIVING THINGS AND THEIR
Oct 10 2019 pdf>. MsAWeems. “3.4 Biogeochemical Cycles Quiz.” ProProfs Quiz Maker
 GEOGRAPHY 100: Human Impacts on the Physical Environment
GEOGRAPHY 100: Human Impacts on the Physical Environment
7-Feb Biogeochemical Cycles: Carbon. Ch. 2 p. 29-34; Ch. 3 p. 65-69. Lab 3: Carbon. Cycle. Quiz 2. T 12-Feb Biogeochemical Cycles: Nitrogen & Phosphorus. Ch. 3
 ERTH/ENVS 3601.03 – Global Biogeochemical Cycles Winter 2020
ERTH/ENVS 3601.03 – Global Biogeochemical Cycles Winter 2020
(available in PDF format in Dalhousie Libraries online). COMMUNICATION. - The Quiz #11 on Chapter 10. Chapter 13 and 14. 23. W12 Thurs Apr 2. Perspectives ...
 Biogeochemical Cycles
Biogeochemical Cycles
in complex cycles called biogeochemical cycles or nutrient cycles. Car- bon
 Lesson 4: The Biogeochemical Cycle
Lesson 4: The Biogeochemical Cycle
Lesson 4 introduces the concept of biogeochemical cycles Mock Bowl Quiz. Standards. Addressed ... Cards.pdf) within their designated spheres.
 Lesson 4: The Biogeochemical Cycle
Lesson 4: The Biogeochemical Cycle
Lesson 4 introduces the concept of biogeochemical cycles Mock Bowl Quiz. Standards. Addressed ... Cards.pdf) within their designated spheres.
 Cycles worksheet
Cycles worksheet
Cycles worksheet. Please answer the following using the words in the text box. Carbon Cycle. 1. Plants use CO2 in the process of
 Biogeochemical Cycles
Biogeochemical Cycles
argument of the necessity of biogeochemical cycles (hydrologic nitrogen
 Carbon and Other Biogeochemical Cycles
Carbon and Other Biogeochemical Cycles
This chapter addresses the biogeochemical cycles of carbon dioxide (http://www.globalcarbonproject.org/global/pdf/DynamicVegetationModels.pdf).
 BIOGEOCHEMICAL CYCLES
BIOGEOCHEMICAL CYCLES
NITROGEN FIXATION__ Process in which nitrogen gas from the atmosphere is converted into ammonia by bacteria that live in the soil and on the roots of plants
 Lesson 5: The Dead Zone
Lesson 5: The Dead Zone
Ask students to recall the preceding lesson on biogeochemical cycles Try to leave enough time before the bowl quiz to go over the answers as a class.
 Carbon Cycle and Energy Flow Quiz Review
Carbon Cycle and Energy Flow Quiz Review
Carbon Cycle and Energy Flow Quiz Review. Quiz Date: ______. This quiz will consist of multiple choice diagraming
 The Carbon Cycle and Atmospheric Carbon Dioxide
The Carbon Cycle and Atmospheric Carbon Dioxide
Responses of the carbon cycle to changing CO2 concentrations. • Uptake of anthropogenic CO2 of biogeochemical parameters in the sea including measure-.
 Lesson 4: National Science The Biogeochemical Cycle
Lesson 4: National Science The Biogeochemical Cycle
The biogeochemical cycle refers to the movement of elements and compounds moving continuously between Earth and its organisms The biogeochemical cycle involves the movement of elements and compounds among four major systems: (1) land and soil (lithosphere) (2) organisms (biosphere) (3) air (atmosphere) and (4) the ocean (hydrosphere)
 BIOGEOCHEMICAL CYCLES Name KEY Name the 4 biogeochemical
BIOGEOCHEMICAL CYCLES Name KEY Name the 4 biogeochemical
Name the 4 biogeochemical cycles you learned about: _____CARBON_____ _____NITROGEN _____ _____PHOSPHORUS_____ _____WATER (HYDROLOGIC) _____ NAME THE CYCLE DESCRIBED: __CARBON_____ Cycle in which photosynthesis and cellular respiration participate
 Searches related to biogeochemical cycles quiz pdf PDF
Searches related to biogeochemical cycles quiz pdf PDF
Biogeochemical Cycles: Reservoirs & Pathways Atmosphere Hydrosphere Lithosphere Biosphere 8 Gaia Hypothesis In the early 1970s James Lovelock theorized that Earth behaves like a superorganism and this concept developed into what is now known as the Gaia hypothesis "Living organisms and their material environment are tightly coupled
What are the different types of biogeochemical cycles?
The most important biogeochemical cycles are the carbon cycle, nitrogen cycle, oxygen cycle, phosphorus cycle, and the water cycle. The biogeochemical cycles always have a state of equilibrium. The state of equilibrium occurs when there is a balance in the cycling of the elements between compartments.
What are the consequences of altered biogeochemical cycles?
Altered biogeochemical cycles together with climate change increase the vulnerability of biodiversity, food security, human health, and water quality to changing climate. However, natural and managed shifts in major biogeochemical cycles can help limit rates of climate change. http://nca2014.globalchange.gov/report/sectors/biogeochemical-cycles
What is the role of microorganisms in biogeochemical cycles?
Microorganisms also require phosphorus for their growth. When the plants and animals die they decompose, and the stored phosphorus is returned to the soil and water bodies which is again consumed by plants and animals and the cycle continues. This biogeochemical cycle moves through the rocks, water bodies and living systems.
Earth's Environmental Systems 83
Consider this: A carbon atom in your ?ngernail today might have helped make up the muscle of a cow a year ago, may have belonged to a blade of grass a month before that, and may have been part of a dinosaur"s tooth 100 million years ago. Matter is never used up, and it never goes away. It just keeps cycling around and around. nutrient Cycling Nutrients cycle through the environment endlessly. Why does matter, such as water, cycle through the environment, never getting used up? Here"s the answer: Matter may be transformed from one type to another, but it cannot be created or destroyed. is principle is called the law of conservation of matter. It explains why the amount of matter in the environment stays the same as it ows through matter cycles, such as the water cycle. Nutrients are matter that organisms require for their life processes. Organisms need several dozen nutrients. e nutrients required in rela- tively large amounts are called macronutrients and include nitrogen, car- bon, and phosphorus. e nutrients needed in small amounts are called micronutrients. Nutrients circulate endlessly throughout the environment in complex cycles called biogeochemical cycles, or nutrient cycles. Car- bon, oxygen, phosphorus, and nitrogen are nutrients that cycle through all of Earth"s spheres and organisms. e water cycle plays parts in all the biogeochemical cycles. the Carbon Cycle Producers play vital roles in the cycling of carbon through the environment. From fossil fuels to DNA, from plastics to medicines, carbon (C) atoms are everywhere. e carbon cycle describes the routes that carbon atoms take through the environment.Biogeochemical Cycles
Guiding Question: How do nutrients cycle through the environment?Explain how the law of conservation of
matter applies to the behavior of nutrients in the environment.Describe the carbon cycle.
Describe the events of the phosphorus cycle.
Explain the importance of bacteria to the nitrogen cycle. Reading StrategyBefore you read, preview Figure 21. Make a list of questions you have about the carbon cycle. As you read, try to answer those questions. Vocabulary law of conservation of matter, nutrient, biogeochemical cycle, primary producer, photosynthesis, consumer, decomposer, cellular respiration, eutrophication, nitrogen xationLESSON
4FOCUS Ask students if they are
familiar with the words nutrition and nutrient. Lead students to an understanding that nutrients are matter that organisms need to survive. Then, have students identify where people get nutrients. (People get nutrients from food.) Point out that the nutrients in food are part of natural cycles that move matter through the nonliving and living parts of the environment (including students" bodies).GUIDING QUESTION
3.4 LESSON PLAN PREVIEW
Real World Students explore
their roles in the carbon cycle.Differentiated Instruction
Advanced learners research
biological molecules contain- ing phosphorus.Inquiry Students design a
controlled experiment to test the eect that adding nutrients has on plant growth.3.4 RESOURCES
Paper and Pencil Activity, E?ects of
CO 2 on Plants- $IBQUFS0WFSWJFX1SFTFOUBUJPOAtmosphere
Atmosphere
Ocean-
atmosphere exchangeOcean-
atmosphere exchangeRunoRuno
Oceans
Fossil fuel
miningFossil fuel
miningDecompositionDecomposition
Soil and
soil organisms Human sources Land plantsConsumers
Sedimentary rock
Oceans
Volcanoes
and other geological processesVolcanoes
and other geological processesBurning of
Fossil fuels
Burning of
Fossil fuels
Decomposers
Cellular
respirationPhotosynthesis
Cellular
respirationCellular
respirationCellular
respirationCellular
respirationCellular
respirationCellular
respirationCellular
respirationPhotosynthesis
ProducersConsumers
WeatheringWeathering
PhotosynthesisPhotosynthesis
Adapted from Schlesinger, W.H. 1997. Biogeochemistry: An analysis of global change, 2nd ed. London: Academic Press
Fossil fuels
Rivers
84 Lesson 4
Producers Primary producers are organisms, including plants and algae, that produce their own food. Producers use the sun"s energy or chemical energy along with carbon dioxide to produce carbohydrates. e carbohydrates are used as food by the producers. Most producers make their own food by using the sun"s energy in photosynthesis. In photosynthesis, producers pull carbon dioxide out of their environment and combine it with water in the presence of sunlight. e chemical bonds in carbon dioxide (CO 2 ) and water (H 2O) are then
broken, producing oxygen (O 2 ) and carbohydrates (such as glucose, C 6 H 12 O 6 ). (e numbers in front of the chemical formulas below are numbers of molecules.) 6CO 2 + 6H 2O + the sun"s energy C
6 H 12 O 6 (sugar) + 6O 2 Consumers and Decomposers ?e carbon in a producer may be passed on to another organism, either a consumer that eats it or a decom- poser that breaks down its wastes or remains. Consumers are organ- isms, mainly animals, that must eat other organisms to obtain nutrients. Decomposers are organisms such as bacteria and fungi that break down wastes and dead organisms.FIGURE 21 Carbon Cycle The carbon
cycle describes the routes that carbon atoms take as they move through the environment. In this diagram, labels in boxes refer to reservoirs, or pools, of carbon, and italic labels refer to processes of the carbon cycle.Earth's Environmental Systems 85
Cellular Respiration Cellular respiration does refer to - ing. Cellular respiration is the process by which organisms use oxygen to release the chemical energy of sugars and release CO 2 and water. In general terms, it is the chemical reverse of photosynthesis. C 6 H 12 O 6 (sugar) + 6O 2 6CO 2 + 6H 2O + energy
Most organisms undergo cellular respiration constantly, releasing carbon back into the atmosphere and oceans. Organisms do not release all the carbon they take in, however. ey use some of it for their life processes. In fact, the abundance of plants and the fact that they use so much carbon for photosynthesis and other processes makes them a major carbon (A sink is a reservoir of a substance that accepts more of that substance than it releases.) Carbon in Sediments When organisms die in water, their remains may settle in sediments. As new layers of sediment accumulate, pres- sure on earlier layers increases. ese conditions can convert so tissues into fossil fuels, and shells and skeletons into sedimentary rock such as limestone. Limestone and other sedimentary rock make up the largest reservoir of carbon. Sedimentary rock releases some of its carbon through erosion and volcanic eruptions. Fossil fuel deposits contain a great deal of carbon, which is released when we extract the fossil fuels.Carbon in Oceans ?e world's oceans are the
second-largest carbon reservoir (Figure 22). ?ey absorb carbon compounds from the atmosphere, from runo, from undersea volcanoes, and from the wastes and remains of organisms. e rates at which seawater absorbs and releases carbon depend on many factors, including the water temperature and the numbers of marine organisms living in it.Human Impacts Humans shi? carbon to the
atmosphere in many ways. By extracting fossil fuels, we remove carbon from storage in the lithosphere. By then burning those fossil fuels, we move carbon diox- ide into the atmosphere. e cutting of forests and burning of forests to plant farm elds also increase carbon in the atmosphere, both by releasing it from storage in plants and by reducing the plants available to use it. Producers cannot absorb enough carbon to keep up with human activities. The Missing Carbon Sink Our understanding of the carbon cycle is not complete. Scientists have long been baed by a missing carbon sink. Of the carbon dioxide humans release, scientists have measured how much is returned to the atmosphere and oceans, and more than1-2 billion metric tons (1.1-2.2 billion tons) are unaccounted for. Many
researchers think it must be taken up by the northern forests. But they"dquotesdbs_dbs11.pdfusesText_17[PDF] Biogeochemical cycles study guide
[PDF] Biogeochemical cycles test answer key
[PDF] biografia de blaise pascal 1623 a 1662
[PDF] biographies definition
[PDF] biographies for 2nd graders
[PDF] Biographies for kids
[PDF] biographies for middle schoolers
[PDF] biographies for teens
[PDF] biographies meaning
[PDF] biographies of women
[PDF] biographies on netflix
[PDF] biographies to read
[PDF] Biographies to read before you die
[PDF] Biography PDF
