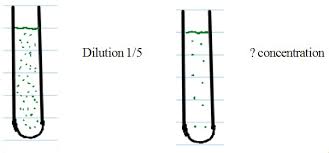
 Dilutions Help
Dilutions Help
CALCULATING THE CONCENTRATION. To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor . CALCULATING THE
 Dilutions Occasionally a solution is too concentrated to be used as it
Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor this by
 Appendix 8: Dilution Factor and Effluent Limitation Calculations for
Appendix 8: Dilution Factor and Effluent Limitation Calculations for
A DF for sites that discharge to freshwater receiving waters in Massachusetts is calculated using the equation below. Alternate calculation methods for DFs may
 PREPARING SOLUTIONS AND MAKING DILUTIONS
PREPARING SOLUTIONS AND MAKING DILUTIONS
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working. (source solution
 Appendix V: Dilution Factor and Effluent Limitation Calculations for
Appendix V: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: Dilution Factor projected downstream concentration of a parameter calculated in accordance with C.
 Preparation and Sequencing of RNA Libraries on the Ion PGM
Preparation and Sequencing of RNA Libraries on the Ion PGM
17-May-2012 After determining the molar concentration of the library calculate the Template Dilution Factor to dilute the library for preparation of ...
 ICH guideline M10 on bioanalytical method validation and study
ICH guideline M10 on bioanalytical method validation and study
25-Jul-2022 At least 5 replicates per dilution factor should be tested in one run to determine if concentrations are accurately and precisely measured ...
 Calculating Nucleic Acid or Protein Concentration Using the GloMax
Calculating Nucleic Acid or Protein Concentration Using the GloMax
07-Aug-2009 Multiply this number by the DNA or RNA constant from Table 1 c. Multiply by the sample dilution factor. 3. Estimate nucleic acid purity a.
 Appendix V: Dilution Factor and Effluent Limitation Calculations for
Appendix V: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: QS + above and the discharge concentration are greater than the WQC calculated for that parameter in.
 Dilutions Help
Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor . CALCULATING THE UNDILUTED CONCENTRATION. Often we wish
 Dilutions Occasionally a solution is too concentrated to be used as it
Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor
 Laboratory Math II: Solutions and Dilutions
Laboratory Math II: Solutions and Dilutions
The concentration of a solution is how much of the solute You will also want to determine your dilution factor. The dilution factor is the factor by.
 1. dilutions and concentrations. liquid and solid samples
1. dilutions and concentrations. liquid and solid samples
of this 1:10 dilution 1 ml corresponds to the sample. properly apply the dilution or concentration factors to determine the microbial density of a ...
 Appendix VI: Dilution Factor and Effluent Limitation Calculations for
Appendix VI: Dilution Factor and Effluent Limitation Calculations for
The equation used to calculate the dilution factor is: downstream concentration of a parameter calculated in accordance with C.1 below
 Groundwater risk assessments for infiltration systems: calculations
Groundwater risk assessments for infiltration systems: calculations
Calculate the dilution factor (DF) for groundwater below the drainage field: 1. Calculate DF taking background concentration into account.
 Determination of Dilution Factors for Discharge of Aluminum
Determination of Dilution Factors for Discharge of Aluminum
flow-ratio DF for streams defined at low flow (7Q10)
 JT - Dilution Factor
JT - Dilution Factor
Dilution Factor: reading gave a concentration of 24.0 ppm for this diluted sample what is the concentration of lead in the original sample? Solution:.
 Appendix V: Dilution Factor and Effluent Limitation Calculations for
Appendix V: Dilution Factor and Effluent Limitation Calculations for
Calculate WQBEL: The freshwater WQBEL is calculated by rearranging the above mass balance equation to solve for the effluent concentration (Cd) by setting the
 Preparation of Solutions Preparation of Solutions
Preparation of Solutions Preparation of Solutions
Oct 17 2016 To make a dilute solution without calculating concentrations use a dilution factor. • Divide the final volume by the initial volume.
 [PDF] Dilution Factor:
[PDF] Dilution Factor:
Dilution factor is defined as: total volume of solution per aliquot volume Where total volume of solution is: 10 0 + 240 0 = 250 0 mL (volumetric flask ) 25
 [PDF] formula 3 Serial Dilutions
[PDF] formula 3 Serial Dilutions
A)Calculate the concentration of the three diluted solutions? ? First: find the D F: Dilution factor (D F) = final volume / aliquot volume
 [PDF] 1 dilutions and concentrations liquid and solid samples
[PDF] 1 dilutions and concentrations liquid and solid samples
We must know not only how to dilute or concentrate a sample; we must also understand how to properly apply the dilution or concentration factors to determine
 [PDF] PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
[PDF] PREPARING SOLUTIONS AND MAKING DILUTIONS - MGEL
The formula below is a quick approach to calculating such dilutions where: V = volume C = concentration; in whatever units you are working (source solution
 [PDF] Dilution-Notespdf - SJU WordPress Sites
[PDF] Dilution-Notespdf - SJU WordPress Sites
Serial dilutions are successive dilutions of a sample The final dilution factor is the multiplicative product of all dilutions 1 mL 1 mL
 [PDF] Laboratory Math II: Solutions and Dilutions
[PDF] Laboratory Math II: Solutions and Dilutions
The concentration of a solution is how much of the solute You will also want to determine your dilution factor The dilution factor is the factor by
 [PDF] Dilutions - Weber State University
[PDF] Dilutions - Weber State University
Since dilutions are simply changes in the ratio of solute to solvent a simple ratio of concentration to volume can be used for almost all calculations
 [PDF] Dilutions Occasionally a solution is too concentrated to be used as it
[PDF] Dilutions Occasionally a solution is too concentrated to be used as it
A general rule to use in calculating the concentration of solutions in a series is to multiply the original concentration by the first dilution factor
 [PDF] Dilutions Help
[PDF] Dilutions Help
To calculate the concentration of our diluted sample we multiply by the inverse of our dilution factor CALCULATING THE UNDILUTED CONCENTRATION Often we wish
 [PDF] How to Make Simple Solutions and Dilutions
[PDF] How to Make Simple Solutions and Dilutions
l Simplc Dilution (Dilution Factor Nlethod based on ratios) per volume and can lrc simply calculated as the concentration t tolune needed = nass of
How do you find the dilution factor based on concentration?
The dilution factor is the inverse of the concentration factor. For example, if you take 1 part of a sample and add 9 parts of water (solvent), then you have made a 1:10 dilution; this has a concentration of 1/10th (0.1) of the original and a dilution factor of 10.How do you calculate the dilution factor?
The formula for dilution factor (or DF for short) is as follows: DF = (final volume of cells + stain)/(initial volume of cells). For example, If you mix your sample 1:1 with AO/PI, you'll need to add 20 uL AO/PI to 20 uL cells, for a total of 40 uL. So, DF = ( 40 uL)/(20uL cells) = 2.What is the dilution factor if you add 0.1 ml aliquot of specimen to 9.9 ml of diluent?
What is the dilution factor if you add 0.1 ml aliquot of a specimen to 9.9 ml of diluent? The dilution factor is equal to the final volume divided by the aliquot volume: 10 mL/0.1 mL = 1:100 dilution.- What is the dilution factor when 0.2 ml is added to 3.8 ml diluent? 4.0/0.2 = 1:20 dilution.
[PDF] how to calculate epinephrine dose
[PDF] how to calculate exchange rate
[PDF] how to calculate experimental yield
[PDF] how to calculate february days
[PDF] how to calculate floor area
[PDF] how to calculate freight cost
[PDF] how to calculate frequency spectrum
[PDF] how to calculate infusion time with drop factor
[PDF] how to calculate infusion time with tubing factor
[PDF] how to calculate logical address in 8086
[PDF] how to calculate mortgage payment on ba ii plus
[PDF] how to calculate multiple regression by hand
[PDF] how to calculate p value from chi square by hand
[PDF] how to calculate physical address in 8086
