 HW # 1: Solution Name ROW ____ PD ____
HW # 1: Solution Name ROW ____ PD ____
Feb 28 2017 A) a compound. B) an element. C) a mixture. D) a substance. 7. Bronze contains 90 to 95 percent copper and 5 to ...
 Heterogeneous Mixture - Homogeneous Mixture Worksheet-Answer
Heterogeneous Mixture - Homogeneous Mixture Worksheet-Answer
Describe the following as an element a compound
 2020-09-14 16:50
2020-09-14 16:50
Sep 14 2020 Classify each of the following as an element
 We use materials every single day – our cotton sheets the springs
We use materials every single day – our cotton sheets the springs
All the properties of elements compounds and mixtures can be classified into two different types: chemical properties and physical properties. Physical
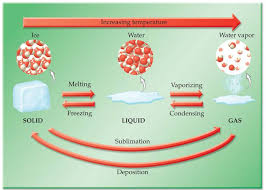 EXAMPLE EXERCISE 4.1 Change of Physical State
EXAMPLE EXERCISE 4.1 Change of Physical State
(b) Copper oxide is a compound of the elements copper and oxygen. (c) Malachite ore is a heterogeneous mixture of copper and other substances. (d) Bronze alloy
 Untitled
Untitled
If the material is a pure substance further classify it as either an element or compound in the right column. or a heterogeneous mixture. 1. Sand.
 Untitled
Untitled
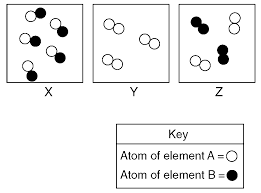
Classify each diagram as: pure substance - element pure substance - compound
 CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
Element. Compound Mixture lead metal table salt. (NaCl). Kool-Aid drink vegetable soup oxygen gas distilled water. Concrete pure gold brass metal flat 7-Up soda.
 Elements Compounds & Mixtures Worksheet
Elements Compounds & Mixtures Worksheet
Elements: •. A pure substance containing only one kind of atom. An element is always uniform all the way through (homogeneous). An element cannot nuclear
 Chemistry: Classifying Matter Name______________________
Chemistry: Classifying Matter Name______________________
If the material is a pure substance further classify it as either an element or compound in the right column. heterogeneous mixture? Give an example of each ...
 EXAMPLE EXERCISE 4.1 Change of Physical State
EXAMPLE EXERCISE 4.1 Change of Physical State
(b) Copper oxide is a compound of the elements copper and oxygen. (c) Malachite ore is a heterogeneous mixture of copper and other substances. (d) Bronze
 Elements Compounds & Mixtures Worksheet
Elements Compounds & Mixtures Worksheet
Elements: • A pure substance containing only one kind of atom. An element is always uniform all the way through (homogeneous)
 Untitled
Untitled
matter including classification as elements compounds and mixtures
 Chemistry: Classifying Matter Name______________________
Chemistry: Classifying Matter Name______________________
substance or a mixture. If the material is a pure substance further classify it as either an element or compound in the right column.
 CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
PURE SUBSTANCES VS. MIXTURES. Classify the following as pure substances (element or compound) or mixtures. 1. sodium Pure substance (E). 11
 Untitled
Untitled
Brass Alloy (Cu + Zn). 10. Oil-Vinegar Elements Compounds Homogeneous Heterogeneme ... Elements. MIXTURES. Heterog. Mix. Homos. Mix. Element. Compound-.
 Whats the Difference? - Atom Element
Whats the Difference? - Atom Element
Compound
 Tiered Activity: Elements Compounds
Tiered Activity: Elements Compounds
http://www.troup.org/userfiles/929/My%20Files/Instructional%20Strategies/tiered_elem_compound_mixtures.pdf?id=14740
 Mixtures Worksheet Answer Key
Mixtures Worksheet Answer Key
If the material is a pure substance identify it as an element or a compound. Material. Mixture. Pure Substance. Homogeneous or Heterogeneous.
 KEY - X Unit 3 Review Packet.pdf
KEY - X Unit 3 Review Packet.pdf
A mixture (is/is not)) a chemical combining of substances. 2. In a compound the (atoms/molecules) are (chemically/physically) combined so that the elements
 Is bronze a mixture or compound? How do you learn the difference?
Is bronze a mixture or compound? How do you learn the difference?
Bronze is a mixture of tin and copper They are heated to the melt point of the higher melting metal I believe and then the other is added
 Is Bronze an Element Compound or Mixture? [ANSWERED]
Is Bronze an Element Compound or Mixture? [ANSWERED]
Bronze is a mixture since it's made of two different elements (Copper and Tin) that combine without forming chemical bond Thus bronze cannot be considered a
 [PDF] Elements Compounds and Mixtures
[PDF] Elements Compounds and Mixtures
An element contains just one type of atom A compound contains two or more different atoms joined together A mixture contains two or more different
 [PDF] Elements Compounds and Mixtures
[PDF] Elements Compounds and Mixtures
An element contains just one type of atom A compound contains two or more different atoms joined together A mixture contains two or more different
 [PDF] MIXTURES ELEMENTS AND COMPOUNDS - Junta de Andalucia
[PDF] MIXTURES ELEMENTS AND COMPOUNDS - Junta de Andalucia
Steel is a homogeneous mixture however it is made from iron and carbon A pure substance is different from a homo- geneous mixture because a pure substance has
 [PDF] ELEMENTS COMPOUNDS & MIXTURES
[PDF] ELEMENTS COMPOUNDS & MIXTURES
A compound is a pure substance that consists of atoms of two or more elements joined together Compounds are formed when atoms of different elements react
 [PDF] Elements Compounds & Mixtures Worksheet
[PDF] Elements Compounds & Mixtures Worksheet
Elements Compounds Mixtures Worksheet Part 1: Read the following information on elements compounds and mixtures Fill in the blanks where necessary
 [PDF] Elements Compounds & Mixtures Notes & Practice
[PDF] Elements Compounds & Mixtures Notes & Practice
%2520compounds
 [PDF] Elements Compounds Mixtures
[PDF] Elements Compounds Mixtures
1 fév 2016 · a mixture containing a metallic element and a non-metallic element • Common alloys include ? Bronze – copper (88 ) and tin (12 )
Is a bronze an element compound or mixture?
Bronze is a mixture of two elements, copper and tin.Is bronze a element or not?
Bronze is a metal alloy that primarily contains copper and 12% tin. Other elements—such as aluminum, arsenic, manganese, phosphorus, and silicon—are also added to yield different properties. These mixtures form some of the common bronze alloys, including: Leaded bronze.Is bronze a mixture and not compound?
Brass and bronze are mixtures.- Bronze is an alloy consisting primarily of copper, commonly with about 12–12.5% tin and often with the addition of other metals (including aluminium, manganese, nickel, or zinc) and sometimes non-metals, such as phosphorus, or metalloids such as arsenic or silicon.