 MODULE 5: DISTILLATION
MODULE 5: DISTILLATION
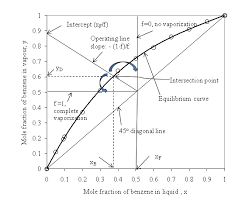
The Figure 5.1 shows a constant pressure phase diagram for an ideal solution. (one that obeys Raoult's Law). At constant pressure depending on relative. Page
 DETERMINATION OF PROLINE IN MIXTURES CONTAINING l-AND
DETERMINATION OF PROLINE IN MIXTURES CONTAINING l-AND
e hodanila$e. III ppt. per cent. 97.8. 93.9. 88.7. 74.4. 10.6 l Meyer (2) has found that this simple equation which follows from Raoult's law
 STUDENTS SUPPORT MATERIAL CLASS-XII CHEMISTRY
STUDENTS SUPPORT MATERIAL CLASS-XII CHEMISTRY
State the following with one application of each: Henry law and Reverse osmosis. State Raoult's law for a solution containing a non volatile solute. Also ...
 Class - XII Multiple Choice Question Bank [MCQ ] Term – I & Term-II
Class - XII Multiple Choice Question Bank [MCQ ] Term – I & Term-II
Redox reactions EMF of a cell
 Properties of Solutions
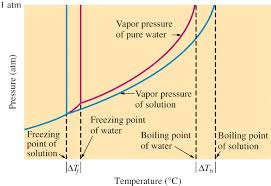
Properties of Solutions
If the mole fraction of heptane is 0.600 what is the composition of the vapor above the solution? Strategy: DOWNSTAIRS use Raoult's law to find vapor pressure
 IB - 1 to 28 - FINAL
IB - 1 to 28 - FINAL
Please shade your mode of examination in the appropriate box PPT or CBT. (A) Point No. 1 of the Application Form: (NAME OF THE CANDIDATE). 19.4. Candidate
 chemistry/xii-(2020-21)
chemistry/xii-(2020-21)
APPLICATION OF HENRY'S LAW. 1. to increase the solubility of carbon dioxide in What is the similarity between Raoult's law and Henry law. Ans. The partial ...
 An Introduction To Headspace Sampling In Gas Chromatography
An Introduction To Headspace Sampling In Gas Chromatography
Raoult's Law states that the vapor pressure of a compound above a solution Figure 22 shows the result of using a ZDL for a typical HS application.
 SCHOOL NAME: WORKSHEET – 7/Module-7(OVERALL) Sub
SCHOOL NAME: WORKSHEET – 7/Module-7(OVERALL) Sub
higher boiling point. 7. Liquids A and B on mixing produce a warm solution. Which type of deviation from. Raoult's law is shown? 8
 LECTURE NOTES
LECTURE NOTES
Dilute Solutions: Vapour Pressure Raoult's Law
 Raoults Law and Its Application to Sublimation Vapor Pressures of
Raoults Law and Its Application to Sublimation Vapor Pressures of
Raoult's Law and Its Application to Sublimation Vapor Pressures of Mixtures of Polycyclic Aromatic Hydrocarbons. Jillian L. Goldfarb and Eric M. Suuberg*.
 DISTRIBUTION LAW (LIMITATIONS AND APPLICATIONS)
DISTRIBUTION LAW (LIMITATIONS AND APPLICATIONS)
APPLICATION OF DISTRIBUTION LAW. ? There are numerous applications of distribution law in the laboratory as well as in industry. (1) Solvent Extraction-.
 PHYSICAL PHARMACY
PHYSICAL PHARMACY
Ideal Solutions and Raoult's Law. In an ideal solution of two volatile liquids the partial vapor pressure of each volatile constituent is equal to the
 (Microsoft PowerPoint
(Microsoft PowerPoint
Dew point & Bubble point calculation with Raoult's Law. Bubble P : calculate (y Application of Raoult's law to species i requires a value for at the.
 VaporILiquid Equilibrium: Introduction
VaporILiquid Equilibrium: Introduction
Thus the liquid water is regarded as pure and Raoult's law for the water (species 2) becomes y2 P = Ppt. At 298.15 K (25'C) and atmospheric pressure
 GUSTAVO V. BARBOSA-CÁNOVAS
GUSTAVO V. BARBOSA-CÁNOVAS
The application of Raoult's Law to food systems is not practical because of solvent-solute interactions. Page 10. „ Assumes water activity lowering due to.
 Applications of Gas Laws in Anaesthesia delivery system- from
Applications of Gas Laws in Anaesthesia delivery system- from
Raoult's law. Definitions of the gas laws. 1)Boyle's Law: states that at constant temperature(T)the volume(V) of a given mass of a.
 An Introduction To Headspace Sampling In Gas Chromatography
An Introduction To Headspace Sampling In Gas Chromatography
To appreciate the principle let's consider an application that is these deviations from the ideal
 Estimation dénergies de GIBBS de solvatation pour les modèles
Estimation dénergies de GIBBS de solvatation pour les modèles
14 janv. 2020 Article : “Application of the corresponding-state law to the ... RAOULT Grandeur écrite en prenant comme référence un mélange de RAOULT.
 Introduction to Thermodynamics models for process engineering
Introduction to Thermodynamics models for process engineering
Raoult's law case of solvent. (Raoult's law) solute fugacity. Henry's law f1 pure f2 pur f2 ref=f2 pure symmetric reference unsymmetric reference.