Why is the reaction between amines and carboxylic acids important?
The reaction between amines and carboxylic acids to form amides is biologically important. It is through this reaction that amino acids (molecules containing both amine and carboxylic acid substituents) link together in a polymer to form proteins.
How amide hydrolysis occurs?
Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. The conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar.
How does amide ion deprotonate carboxylic acid?
In the last step, the amide ion deprotonates the carboxylic acid to form a carboxylate salt and an amine as products. This final deprotonation step essentially removes the carboxylic acid from the equilibrium which drives the reaction towards completion.

Acid and base-catalyzed hydrolysis of amides Organic chemistry Khan Academy

Nomenclature and properties of amides Organic chemistry Khan Academy

Preparation of amides using DCC Organic chemistry Khan Academy
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the 1° |
|
Mode of Activation of Cobalt(II) Amides for Catalytic Hydrosilylation
Jan 25 2017 that cobalt(II) amides can react with hydrosilane to form cobalt(I) species |
|
Amides Are Novel Protein Modifications Formed by Physiological
Apr 20 2001 ducing sugars |
|
Amide activation: an emerging tool for chemoselective synthesis
Aug 28 2018 catalysis |
|
Durham E-Theses - Direct Amide Formation Between Carboxylic
Carboxylic acids and amines can react together to form salts strong heating of these salts can lead to amide formation |
|
Carboxylic Acid Deoxyfluorination and One-Pot Amide Bond
Jul 26 2021 deoxyfluorination amide bond-forming reaction gives ready access to ... with the ?-fluoroamines |
|
ADVANCED GAS CHROMATOGRAPHY – PROGRESS IN
reagents can be used alone to form esters ethers and amides or they can be used in conjunction with acylation or silylation reagents. The reaction |
|
An efficient procedure for chemoselective amidation from carboxylic
Apart from carboxylic acids amides can also be synthesized initial formation of stable carboxylic acid ammonium salt upon mixing [2]. Such a. |
|
Opinion on Nitrosamines and Secondary Amines in Cosmetic Products
following the ranking of carcinogenic potency of nitrosamines in question? Comment The generated acetaldehyde can react with ethanolamine to form 5-. |
|
Reactions of Amines
Reaction with Ketones or Aldehydes (Section 18-16,17 and 19-10) R' R O aldehyde Must have at least 2 H's on nitrogen → 2º, 3º amines can't do this Rank the following in terms of boiling point, 1 being highest, 4 being lowest OH Add base to the aqueous layer, to convert DH+ back to neutral D In it's neutral form, |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
With heat and an acid catalyst, an amide can be hydrolyzed to produce a carboxylic acid and an amine (or ammonia) A specific example of this reaction is the hydrolysis of N-methylpropanamide |
|
Chapter 17: Amines and Amides
Nitrogen atoms forms three covalent bonds and has the following bonding Nitrogen can form three covalent bonds to complete its octet of electrons in peptides Amides are most usually formed by the reaction of an acid chloride ( R- COCl |
|
Amide Acetals, Ester Aminals, and Ortho Amides - ScienceDirect
Some of these compounds—the amide acetals in particular—are useful synthetic Amide acetals can be divided into two groups: heterocyclic amide acetals, extent of side reactions, principally ether and amide formation due to attack |
|
B(OCH2CF3)3-mediated amidation reactions - UCL Discovery
procedure was developed which enables the purification of all of these amide as this anhydride 14 can react with the amine to form the amide product |
|
PHYSICOCHEMICAL PROPERTIES OF ORGANIC MEDICINAL
Like amines, amides can be classified as "primary", "secondary" or "tertiary" depending on the Show the products formed in the following reactions: O N H H |
|
[PDF] Chapter 6 Amines and Amides - Angelo State University
Learn the major chemical reactions of amines and amides, and learn how to Provide common names for the following 2° and 3° 1° and 2° amines can hydrogen bond to each other N Amides are formed when acid chlorides react with |
|
[PDF] Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
3) Bond the nitrogen and its remaining groups to the carbonyl carbon of the carboxylic acid An amide can form when a carboxylic acid reacts with ammonia, with a primary, or with a secondary amine Examples a) Formation of an amide by the reaction of a carboxylic acid and ammonia |
|
[PDF] Reactions of Chapter 9 Summary, Worksheet, and Key Summary of
3) Formation of Amides The Reaction of Carboxylic Acids with Amines • An amide is With heat and an acid catalyst, an amide can be hydrolyzed to produce a carboxylic acid and an amine (or 7) Complete the following reactions a) O |
|
[PDF] Synthesis and biological importance of amide - Pulsus Group
he Amide bond formation reactions are among the most important transformations in However, these classical approaches are low in atom efficiency and alcohols or amines, can be now conveniently employed as starting materials for the |
|
[PDF] Reactions of Amines
this time) • Must have at least 2 H's on nitrogen → 2º, 3º amines can't do this Biologically amine + acid → amide is routine, and is facilitated by complex enzyme mechanisms 5 Exactly as for amide formation • Many antibiotic Synthesis of Amines Draw the products for the following reactions 53 O NaBH3CN, H+ |
|
Amides Are Novel Protein Modifications Formed by Physiological
Apr 20, 2001 · links derived from the reaction of lysine with glyoxal and GOLA in human lens proteins shows that these amides can form in human tissue |
|
Amide Acetals, Ester Aminals, and Ortho Amides - ScienceDirectcom
These include the amide acetals (1), ester aminals (2), the ortho amides (3), and Amide acetals can be divided into two groups heterocyclic amide acetals, extent of side reactions, principally ether and amide formation due to attack |
- amines and amides worksheet with answers
- reactions of amides
- chemical properties of amides
- reactions of amines pdf
- amine reaction with water
- amine + naoh
- carboxylic acid
- what do amides smell like
- amides can be formed by the reaction of which of the following
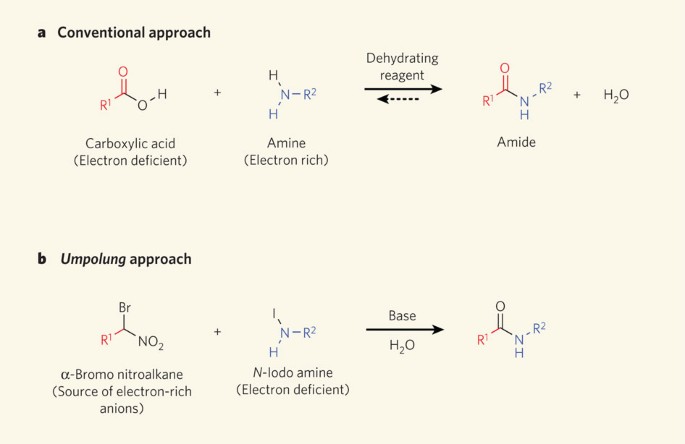
The Amide Functional Group: Properties Synthesis and Nomenclature
Source:https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2F4651020a/MediaObjects/41586_2010_Article_BF4651020a_Fig1_HTML.jpg
Amide bonds made in reverse
Source: Nature

The Amide Functional Group: Properties Synthesis and Nomenclature
Source:https://pubs.rsc.org/image/article/2020/sc/d0sc01330g/d0sc01330g-f1_hi-res.gif
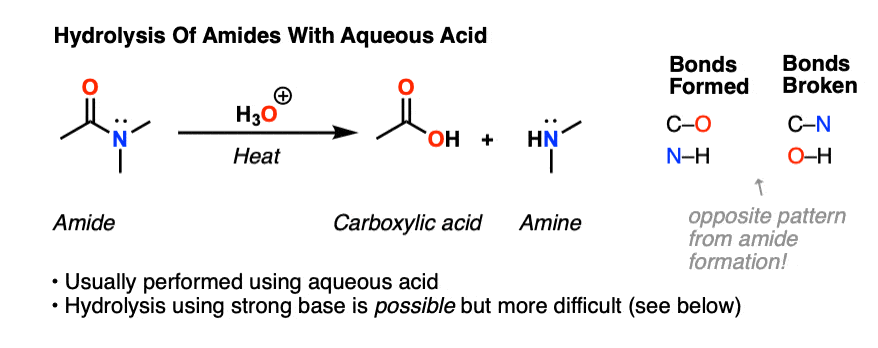
Synthesis of secondary and tertiary amides without coupling agents
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/10/2-opposite-of-condensation-is-hydrolysis-which-can-be-achieved-through-aqueous-acid-treatment-of-amides-or-concentrated-hcl-to-give-carboxylic-acid-and-amine.gif
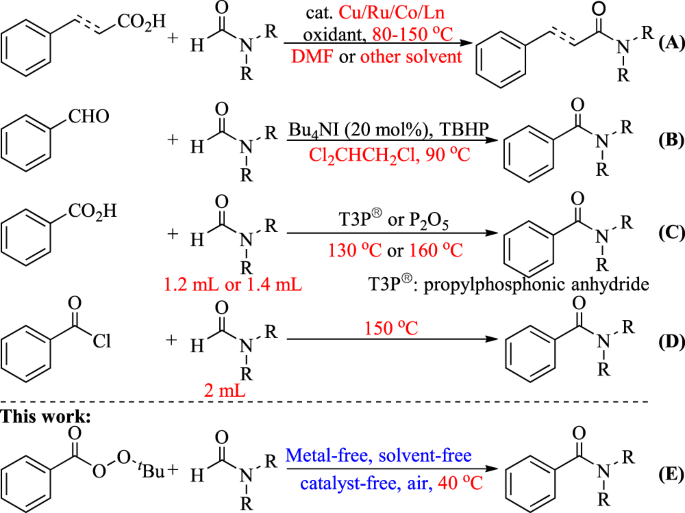
Amide Hydrolysis Using Acid Or Base – Master Organic Chemistry
Source:https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41598-019-39240-z/MediaObjects/41598_2019_39240_Fig1_HTML.png

Metal- and solvent-free synthesis of amides using substitute
Source:https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/3141184c-47b2-4a33-8096-c180e69f58fc/mscheme1.jpg
amine + hcl mechanism
[PDF] 610 Amines - chemrevise
- amine reaction with hcl
- amine reaction with water
- imine to amine mechanism
- reaction of tertiary amine with hcl
- reaction of amines with nitrous acid mechanism
- amine reactions
- amines mechanism
- amine examples
- amine + hcl mechanism
amine + koh
[PDF] Desiccant efficiency in solvent and reagent drying 5 Amines
- hydroxyl reaction with amine
- alcohol and amine reaction
- amine examples
- amine condensation
- amine reaction with acid
- amine to cn
- amine amine coupling
- reactions involving amines
- amine + koh
- amine koh reaction
- koh amine test
- amine odor koh poc
- amine value koh
- amine value mg koh/g
- amine number mg koh/g
- amine kohen
amine acetic acid reaction
THE REACTION OF IODOACETIC ACID ON MERCAPTANS AND
- reactions of amines pdf
- amine reaction with water
- amine reaction with hcl
- amine reaction with acid
- amine reaction with hydroxyl group
- amine + hcl
- amine + naoh
- amine reactions practice problems
- acetic acid and methylamine reaction
amine acetylation mechanism
An Efficient Method for Acylation Reactions - ScienceDirectcom
- acetylation of amines with acetic anhydride mechanism
- acetylation of primary amine
- acetylation of alcohol mechanism
- acetylation reaction mechanism
- acetylation mechanism acetic anhydride
- acetylation of phenol mechanism
- acetylation reaction of amines
- acetylation of alcohol with acetic anhydride mechanism