How do aliphatic amines react with nitrous acid?
Primary aliphatic amines with nitrous acid give very unstable diazonium salts which spontaneously decompose by losing N 2 to form a carbenium ion. The carbenium ion goes on to produce a mixture of alkenes, alkanols or alkyl halides, with alkanols as the major product.
How do amines form salts?
Amine - Reactions, Synthesis, Properties: Amines characteristically form salts with acids; a hydrogen ion, H+, adds to the nitrogen. With the strong mineral acids (e.g., H2SO4, HNO3, and HCl), the reaction is vigorous. Salt formation is instantly reversed by strong bases such as NaOH.
How do amines react with acid chlorides?
Ammonia, 1 o amines, and 2 o amines react rapidly with acid chlorides or acid anhydrides to form 1 o, 2 o, and 3 o amides respectively (Sections 21-4 and 21.5). These reactions typically take place rapidly at room temperature and provide high reaction yields.
What happens if amine salt is heated to 200°C?
Upon heating to 200°C, the primary and secondary amine salts dehydrate to form the corresponding amides. Amines R 3 N react with strong acids such as hydroiodic acid (HI), hydrobromic acid (HBr) and hydrochloric acid (HCl) to give ammonium salts R 3 NH +. Nitrous acid with the chemical formula HNO 2 is unstable.

Acid and base-catalyzed hydrolysis of amides Organic chemistry Khan Academy

Amine Synthesis Reactions

Amine Synthesis Reactions Organic Chemistry
|
Reactions of Amines
amine base ammonium salt. (acidic). • Mechanism: Required (protonation) Some base is required for the deprotonation step and to absorb the HCl. |
|
Kinetics and Mechanism of N-Boc Cleavage: Evidence of a Second
10 Nov 2010 The kinetics of the HCl-catalyzed deprotection of the Boc-protected amine thioester 2 to liberate. AZD3409 1 have been studied in a mixture ... |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the Complete the following reactions: CH3NH2 + HCl. NHCH3. + HCl. |
|
Mechanism of the Reaction of Imido Esters with Amines
amine hydrochloride was found by titration to contain ap- proximately 5% ammonium chloride after the kinetic experiments were. |
|
CH 2280 Procedure for Metal/Acid Reduction of Nitro Compounds
Proposed mechanism for tin/HCl reduction of nitro compounds. disposable pipet at a time) until the pH is about 10 (this will precipitate the amine. |
|
Mechanism studies on thermal dissociation of tri-n-octylamine
2 Sept 2017 Abstract. The thermal dissociation of tri-n-octylamine hydrochloride (TOAHCl) was investigated using both the quantum chemical simulation ... |
|
Mechanism of the Formation of Guanidine and its Derivatives by the
Fusion of Dicyandiamide with Amine Salt. By Kllchlro SUGINO duct.4) In order to clarify this mechanism ... dicyandiamide and aniline hydrochloride was. |
|
Highly Efficient Oxidative Amidation of Aldehydes with Amine
We rationalized that the corresponding amine hydrochloride salt A tentative mechanism for the oxidative amidation of aldehydes. |
|
Unraveling the Mechanistic Origins of Epoxy Degradation in Acids
20 Jun 2019 behavior of hydrochloric acid (HCl) into an amine-cured ... mechanism of HCl acids into polymers industrial experience. |
|
The Mechanism of Action of Ethanolamine Ammonia-Lyase a B12
amine the conversion of 2-aminopropanol dence in support of the mechanism previously proposed for ... 0.05 M ethanolamine hydrochloride. |
|
Reactions of Amines
mechanisms 5 Substitution for Aromatic Amines via the Diazonium Salts (“The Sandmeyer Reaction”) (Section 19-17, 18) ArNH2 NaNO2, HCl ArN2 Cl |
|
CHAPTER 7 AMINES
Write the equation for the neutralization of trimethylamine with HCl A molecule with two amine groups can react with two molecules of HCl or other acid |
|
311 Amines
Amines are derivatives of ammonia where at least 1 H has been replaced with an R group: Primary Reflux The Mechanism – Nucleophilic substitution Reflux • NaOH is added to release phenylamine from its salt (with the HCl) • This is an |
|
Reaction of glucose with some amines - CORE
Ling and Hanji (81,82) made a study of the mechanism of carameGLization of amine was obtained and was isolated as the hydrochloride They prepared |
|
Reactions of Amines: Reductive Amination (Borch Reaction
When ketone or aldehydes react with 1° amines an imine is formed NH2 / NaBH3CN, HCl (1eq) CH3OHsolvent NH 1) 2) neutralization, NaOH Page 2 The mechanism for the use of a non-‐metal-‐hydride as the reducing agent is |
|
610 Amines - chemrevise
As the reaction is carried out in HCl the salt C6H5NH3 The same reaction mechanism occurs with the secondary amine reacting to form a tertiary amine CH 3 |
|
Experiment 16 Qualitative Analysis of Amines and Amine Unknown
This top layer, however, will be soluble in 5 HCl and this distinguishes tertiary from secondary amines Repeat the Hinsberg test with your unknown Nitrous Acid |
|
[PDF] Reactions of Amines
mechanisms 5 Substitution for Aromatic Amines via the Diazonium Salts (“The Sandmeyer Reaction”) (Section 19 17, 18) ArNH2 NaNO2, HCl ArN2 Cl |
|
[PDF] Reactions of Amines: Reductive Amination (Borch Reaction
When ketone or aldehydes react with 1° amines an imine is formed NH2 NaBH3CN, HCl (1eq) CH3OHsolvent NH 1) 2) neutralization, NaOH Page 2 The mechanism for the use of a non ‐metal ‐hydride as the reducing agent is |
|
[PDF] 311 Amines - chemhumecouk
Conditions Reflux The Mechanism – Nucleophilic substitution NaOH is added to release phenylamine from its salt (with the HCl) • This is an important |
|
[PDF] Amines Amines are organic compounds containing a nitrogen
With a saturated amine, the lone pair resides in a sp3 hybridized orbital H3 C Instead of an E2 base mechanism, the Cope occurs through an oxidation mechanism salt is generated from an aniline derivative NH 2 NaNO 2 HCl N N |
|
[PDF] Chapter 19 – Amines
HCl dipropyl amine phenol decanoic acid diethyl ether phenol dipropyl ammonium Cl of aryl diazonium salts ▫ You don't need to know any mechanisms here |
|
[PDF] Coupling of substances containing a primary amine to - DiVA portal
unspecific reaction which is not suited for the coupling of primary amines to HA The mechanism is as follows; the dissociated hydroxyl group of NHS 8 makes a ≥995 (CAS 7647 14 5), hydrochloric acid 12 M (CAS 7647 01 0) 42 |
|
[PDF] 610 Amines - chemrevise
As the reaction is carried out in HCl the salt C6H5NH3 The same reaction mechanism occurs with the secondary amine reacting to form a tertiary amine CH 3 |
- amine reaction with hcl
- amine reaction with water
- imine to amine mechanism
- reaction of tertiary amine with hcl
- reaction of amines with nitrous acid mechanism
- amine reactions
- amines mechanism
- amine examples
- amine + hcl mechanism

Protecting Groups for Amines: Carbamates – Master Organic Chemistry
Source:https://s3-us-west-2.amazonaws.com/courses-images/wp-content/uploads/sites/3773/2018/11/30162750/hofmannrearrangement2.png
232 Preparation of Amines
Source: Organic Chemistry II
PDF) Acetylation of 3-aminopyridine: The Schotten-Baumann Reaction
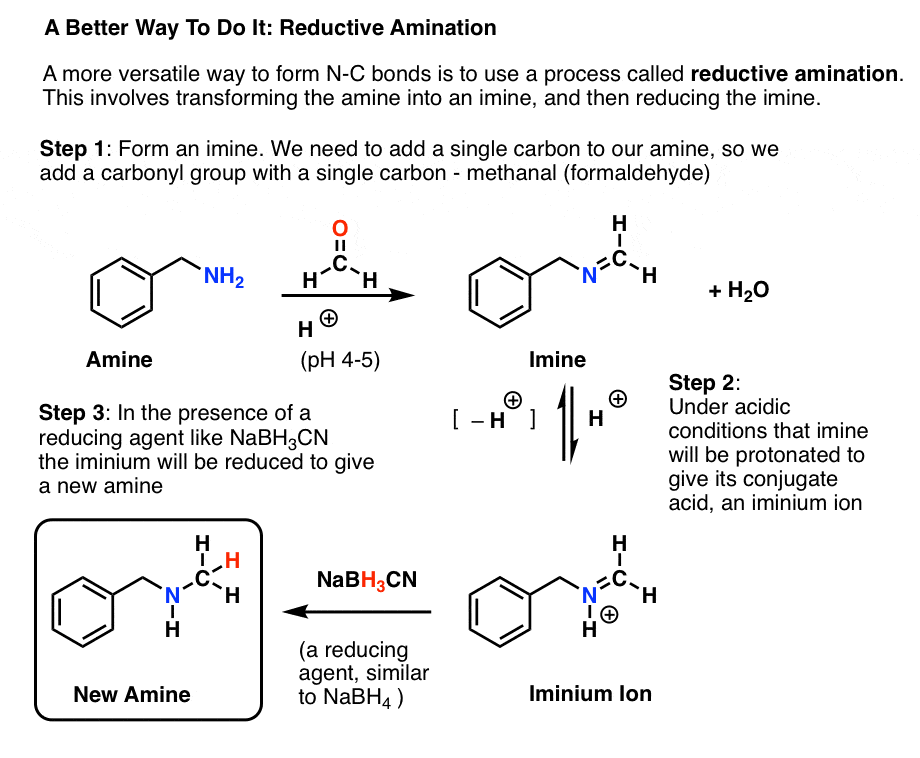
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/3-what-is-reductive-amination-shows-formation-of-imine-from-amine-and-then-reduction-to-give-n-methyl-amine.gif

Reductive Amination and How It Works – Master Organic Chemistry
Source:https://upload.wikimedia.org/wikipedia/commons/thumb/f/f5/Mannichreactionmech1.svg/567px-Mannichreactionmech1.svg.png

Mannich reaction - Wikipedia
Source:https://s3.amazonaws.com/dev-hmhco-vmg-craftcms-public/_cliffsnotes/assets/23180.jpg

Preparation of Amines
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/F1-fate-of-isocyanate-in-hofmann-and-curtius-rearrangement-formation-of-urea-amine-carbamate-mechanisms.gif
amine + koh
[PDF] Desiccant efficiency in solvent and reagent drying 5 Amines
- hydroxyl reaction with amine
- alcohol and amine reaction
- amine examples
- amine condensation
- amine reaction with acid
- amine to cn
- amine amine coupling
- reactions involving amines
- amine + koh
- amine koh reaction
- koh amine test
- amine odor koh poc
- amine value koh
- amine value mg koh/g
- amine number mg koh/g
- amine kohen
amine acetic acid reaction
THE REACTION OF IODOACETIC ACID ON MERCAPTANS AND
- reactions of amines pdf
- amine reaction with water
- amine reaction with hcl
- amine reaction with acid
- amine reaction with hydroxyl group
- amine + hcl
- amine + naoh
- amine reactions practice problems
- acetic acid and methylamine reaction
amine acetylation mechanism
An Efficient Method for Acylation Reactions - ScienceDirectcom
- acetylation of amines with acetic anhydride mechanism
- acetylation of primary amine
- acetylation of alcohol mechanism
- acetylation reaction mechanism
- acetylation mechanism acetic anhydride
- acetylation of phenol mechanism
- acetylation reaction of amines
- acetylation of alcohol with acetic anhydride mechanism
amine alcohol condensation
Amination of diols and polyols to acyclic amines - ScienceDirectcom
- alcohol to amine reaction
- alcohol to amine mechanism
- alcohol amine coupling
- secondary amine reaction with alcohol
- alcohol to amine mitsunobu
- alcohol to tertiary amine
- amine synthesis
- primary alcohol to secondary amine