What is the mechanism of acid hydrolysis?
The nucleophile involved in acid-catalyzed hydrolysis of the ester is H2O.
The water molecule attacks the carbonyl carbon atom by donating the oxygen's lone pair of electrons and forming a bond.
It is a nucleophilic substitution reaction as water act as a nucleophile, by attacking the carbonyl carbon of ester.What is the mechanism of nitrile lialh4?
Nitriles can be converted to 1° amines by reaction with LiAlH4.
During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion.
Once stabilized by a Lewis acid-base complexation the imine salt can accept a second hydride to form a dianion.What is the mechanism of the nitrile reaction?
The mechanism begins with the nucleophilic Grignard reagent reacting with the electrophilic carbon of the nitrile to form a salt of the imine anion.
The imine salt is protonated to form an imine which is subsequently protonated to form a positively charged iminium ion.2 fév. 2024
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
For example a typical reaction of nitriles is addition. The mechanism of nitrile hydrolysis in acidic solution involves |
|
20_18_20.html.ppt [Read-Only]
Hydrolysis of nitriles resembles the hydrolysis In basic solution the carboxylic acid product ... The mechanism of amide formation is analogous. |
|
A Mechanism for the Hydrolysis of Cyanamide in Acid Solution
greater tendency toward formation of IV and V. (or VI) in alcohol than in water. Hydrolysis of Nitriles. The strong acids are as specific in their effect on the |
|
Theoretical study of nitrile hydrolysis by solid acid catalysts
1 janv. 2000 Subject headings: reaction mechanism; hydrolysis/ nitrile/ quantum ... catalyze both acid and base hydrolysis reactions (18). |
|
The von Richter Reaction. V. Evidence that Benzonitrile and
A mechanism in harmony with known facts is presented. for hydrolysis of the nitrile to the acid under these conditions. |
|
Article - Direct conversion of aromatic amides into crystalline
15 nov. 2021 nitrile conversion mechanism exhibited an RDS with higher ... the hydrolysis of nitrile to benzoic acid is shown in Supplemental Fig. 13. |
|
Chapter 21: Carboxylic Acid Derivatives
mechanism it is possible to transform one acid The reactivity of acid derivatives can be correlated ... Base Catalyzed Hydrolysis of Nitriles. |
|
THE HYDROLYSIS OF HYDROGEN CYANIDE BY ACIDS1
Walker and Krieble namely |
|
Carboxylic acid Derivatives
The most important acid derivatives are esters amides and nitriles |
|
Carbanion mechanism of ester hydrolysis. II. o-Nitrophenyl .alpha
oNitrophenyl -Cyano- and -Dimethylsulfonioacetate Esters The second-order rate constant for alkaline hydrolysis of p-nitrophenyl acetate is within. |
|
Hydrolysis of Nitriles
Hydrolysis of nitriles resembles the hydrolysis of amides The reaction is In basic solution the carboxylic acid product Mechanism of Hydrolysis of Nitriles |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
lation between nitriles and carboxylic acid derivatives highlighted by this pair of Bruckner R (author), Harmata M (editor) In: Organic Mechanisms – Reactions, is that the hydrolysis amide → carboxylic acid or amide → carboxylate can only |
|
88 Chapter 20: Carboxylic Acids and Nitriles 201 Naming
Acid or basic hydrolysis of a nitrile (mechanism, Fig 20 4) cyanide ion is an excellent nucleophile and will react with 1° and 2° alkyl halides and tosylates to give nitriles This reaction add one carbon |
|
Theoretical study of nitrile hydrolysis by solid acid catalysts
1 jan 2000 · Subject headings: reaction mechanism; hydrolysis/ nitrile/ quantum chemistry catalyze both acid and base hydrolysis reactions (18) Its active |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
for the hydrolysis of nitriles are more severe The mechanism of nitrile hydrolysis in acidic solution involves, first, protonation of the ni- trogen (Sec 21 5): |
| Hydrolysis of Nitriles |
|
Carboxylic Compounds, Nitriles, and Their Interconversion
D, a so called orthoester, can either be isolated as such or hydrolyzed to give a normal carboxylic acid ester B under mild conditions (mechanism Figure 913) In |
|
[PDF] and nitrile hydrolysis
methods for nitrile hydrolysis to amides use strong acid (96 H2SO4)13 or base (50 SPO complexes is described and a possible mechanism is discussed |
|
[PDF] 88 Chapter 20: Carboxylic Acids and Nitriles 201 Naming
Acid or basic hydrolysis of a nitrile (mechanism, Fig 204) cyanide ion is an excellent nucleophile and will react with 1° and 2° alkyl halides and tosylates to give |
|
[PDF] 217 hydrolysis of carboxylic acid derivatives - Sapling
for the hydrolysis of nitriles are more severe The mechanism of nitrile hydrolysis in acidic solution involves, first, protonation of the ni trogen (Sec 215) |
- basic hydrolysis of nitriles
- nitrile to carboxylic acid
- cyano to acid
- nitrile hydrolysis procedure
- hydrolysis of cyanide in acidic medium
- nitrile to carboxylic acid mechanism
- rcn h3o+
- cn and h3o+
- acidic hydrolysis of nitriles mechanism
- acidic hydrolysis of nitriles
- alkaline hydrolysis of nitriles
- alkaline hydrolysis of nitriles mechanism
- acid hydrolysis of nitriles conditions
- acid hydrolysis of nitrile to amide
- acidic hydrolysis of cyanide
- alkaline hydrolysis of cyanide
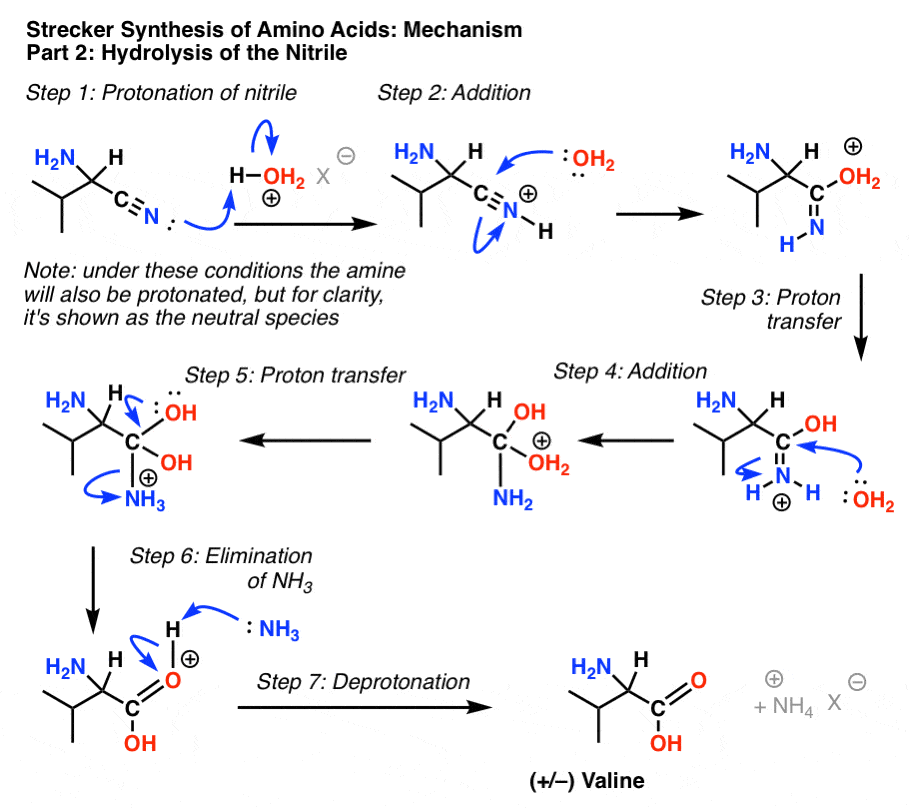
The Mechanism of Nitrile Hydrolysis To Carboxylic Acid - Chemistry
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/5-mechanism-of-strecker-synthesis-part-2-hydrolysis-of-nitrile.gif

The Strecker Synthesis of Amino Acids – Master Organic Chemistry
Source:https://www.chemistrysteps.com/wp-content/uploads/2020/03/Nitrile-hydrolysis.png

The Mechanism of Nitrile Hydrolysis To Carboxylic Acid - Chemistry
Source:https://ars.els-cdn.com/content/image/1-s2.0-S1350417710000726-gr7.jpg

Swift and efficient sono-hydrolysis of nitriles to carboxylic
Source:https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/10/6-hydrolysis-of-amides-mechanism-first-step-is-protonation-of-oxygen-of-amide.gif

Amide Hydrolysis Using Acid Or Base – Master Organic Chemistry
Source:http://www.chem.ucalgary.ca/courses/350/Carey5th/Ch20/amideh3omech.gif

Ch20 : Amide hydrolysis
Source:https://www.chem.ucalgary.ca/courses/351/Carey5th/Ch20/rcnh3omech.gif
acidic strength of benzoic acid derivatives
[PDF] Contribution of Substituent Phenols and Benzoic Acids Groups
- why benzoic acid is more acidic than para hydroxybenzoic acid
- is picric acid more acidic than benzoic acid
- is picric acid more acidic than formic acid
- pka of benzoic acid
- salicylic acid is stronger than benzoic acid
- acidic strength of benzoic acid
- acidic strength of benzoic acid derivatives
- acidic strength of benzoic acid and acetic acid
- acidic strength of benzoic acid and p aminobenzoic acid
- acidic strength of substituted benzoic acids
- acidic strength of halo benzoic acid
- acidic strength of methyl benzoic acid
- acidic strength of methoxy benzoic acid
acidity and basicity of alcohols and phenols
[PDF] physicochemical properties of organic medicinal agents
- alcohol phenol and ether questions pdf
- benzyl alcohol vs phenol acidity
- alcohol phenol and ether reactions
- acidity of phenol
- how are phenols and alcohols similar
- will alcohol phenol soluble in water why
- alcohols and phenols lab report answers
- acidity of alcohols
- acidity and basicity of organic compounds
- acidity and basicity of salts
- acidity and basicity of amines
- acidity and basicity of salt solutions
- acidity and basicity of amino acids
- acidity and basicity of alcohols
- acidity and basicity of organic compounds pdf
- acidity and basicity of aromatic compounds
acidity in drinking water
[PDF] LEVEL OF pH IN DRINKING WATER OF AN OIL AND GAS
- how to raise ph in drinking water
- causes of acidity in water
- how to lower ph in drinking water
- drinking water ph level chart
- alkaline water
- ph of water
- ph of drinking water
- acidity of water
- acidity limit in drinking water
- reduce acidity in drinking water
- total acidity in drinking water
- high acidity in drinking water
- determination of acidity in drinking water
- effects of acidity in drinking water
- causes of acidity in drinking water
- acidity in stomach after drinking water
acidity of alcohols phenols and carboxylic acids
[PDF] Alcohols and Phenols Chem 145
- alcohols and phenols pdf
- acidity of alcohols and phenols
- benzyl alcohol vs phenol acidity
- how are phenols and alcohols similar
- how alcohol differ from phenol
- acidity of phenol
- will alcohol phenol soluble in water why
- alcohol phenol and ether reactions
- acidity of alcohols phenols and carboxylic acids
- acidity of alcohols and phenols
- acidity of alcohols and phenols class 12
- relative acidity of alcohols phenols and carboxylic acids
- acidity of phenol alcohol and water
- difference in acidity of alcohols phenols and carboxylic acids
- compare acidity of alcohols and phenols
- acidity and basicity of alcohols and phenols