 Exercise 15.4 - Titrations - Answers.pdf
Exercise 15.4 - Titrations - Answers.pdf
Solving Titration Problems. A titration is a chemical process for finding the The pH of any strong acid Istrong base titration at the equivalence point.
 ACID-BASE TITRATIONS (PROBLEMS)
ACID-BASE TITRATIONS (PROBLEMS)
being titrated with 0.50 M NaOH. Calculate the pH of this solution initially before any NaOH is added. At this point
 Problem Solving
Problem Solving
Apr 20 2016 Mole ratio of base to acid in titration reaction ? Volume of base solution titrated. 20.00 mL. Moles of base in solution titrated ? mol NaOH.
 Acid-Base Titration and pH
Acid-Base Titration and pH
PROBLEMS Write the answer on the line to the left. Show all your work in the space provided. 4. [H3O ] in an aqueous solution 2.3 10 3 M. 4.3
 Acid – Base Titration Calculations (WA + SB)
Acid – Base Titration Calculations (WA + SB)
Calculate the pH of 50.0 ml of a 0.20 M solution of lactic acid HC3H5O3 after it has been titrated with a total of 30.0 ml of 0.40 M KOH? Page 2. P G4 A (pg of ).
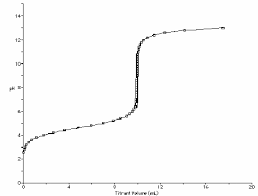 1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
What Kind of Solution/pH at End? p2 Titration Calculations p11. Preparation and Recognition of Buffers p4 pH Estimations/Calculations after acid/base are
 Untitled
Untitled
EXTRA PRACTICE: Titration Problems Practice. Titration Calculate the moles of base used the moles of acid used and the concentration of the original acid.
 Acid – Base Titration Calculations (WB + SA)
Acid – Base Titration Calculations (WB + SA)
1. Calculate the pH of 20.0 ml of a 0.060 M solution of ammonia NH3. 2. What volume of 0.040 M HI would be needed to
 Test3 ch17b Buffer-Titration-Equilibrium Practice Problems
Test3 ch17b Buffer-Titration-Equilibrium Practice Problems
Answer: A buffer consists of a weak acid and its conjugate base in roughly equal amounts. If acid is added to the solution it is consumed by the conjugate
 What is the M of NaOH if it takes 40.0 ml of NaOH to reach the
What is the M of NaOH if it takes 40.0 ml of NaOH to reach the
Worksheet - Titration Problems. 1. What is the M of NaOH if it takes 40.0 ml H₂Soy as Hel to neutralize the base. 30.3 ml of 0.305 M NaOH are required to ...
 w336-titrations-worksheet.pdf
w336-titrations-worksheet.pdf
calculations for the concentration of the base? 3). It takes 38 mL of 0.75 M NaOH solution to completely neutralize 155 mL of a sulfuric acid solution
 Exercise 15.4 - Titrations - Answers.pdf
Exercise 15.4 - Titrations - Answers.pdf
Solving Titration Problems While the volumes of acid and base should probably be converted to liters as long as they are the same unit
 Acid – Base Titration Calculations (WA + SB)
Acid – Base Titration Calculations (WA + SB)
Calculate the pH of 50.0 ml of a 0.20 M solution of lactic acid HC3H5O3 after it has been titrated with a total of 30.0 ml of 0.40 M KOH? Page 2. P G4 A (pg of ).
 Titration Calculations Strong Acid/Strong Base Calculations (1) Use
Titration Calculations Strong Acid/Strong Base Calculations (1) Use
added during a titration to 25.0 mL of a 0.12M HCl solution with 0.15M NaOH solution? For strong acid/base titration perform stoichiometry calculation
 Bookmark File PDF Acid Base Titration Lab 39 Answers ? - covid19
Bookmark File PDF Acid Base Titration Lab 39 Answers ? - covid19
easy to acquire as without difficulty as download lead Acid Base Titration. Lab 39 Answers. It will not say yes many grow old as we accustom before.
 1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
1 General Chemistry II Jasperse Buffers/Titrations/Solubility. Extra
What Kind of Solution/pH at End? p2 Titration Calculations p11. Preparation and Recognition of Buffers p4 pH Estimations/Calculations after acid/base.
 Where To Download Redox Practice Problems With Answers Copy
Where To Download Redox Practice Problems With Answers Copy
with states phases
 A Dating Analogy for Acid-Base Titration Problems
A Dating Analogy for Acid-Base Titration Problems
alleviate this confusion. The use of other analogies to help clarify other confusing concepts related to solution concen- trations and stoichiometric ratios
 Worksheet 22 – Weak Acid/Strong Base Titrations A. Initial pH This
Worksheet 22 – Weak Acid/Strong Base Titrations A. Initial pH This
This can only be used after the initial point and before the equivalence point when the acid and its conjugate base are both major species in the solution. At
 Test3 ch17b Buffer-Titration-Equilibrium Practice Problems
Test3 ch17b Buffer-Titration-Equilibrium Practice Problems
What Kind of Solution/pH at End? p2 Titration Calculations p11. Preparation and Recognition of Buffers p4 pH Estimations/Calculations after acid/base.
Worksheet 22 - Weak Acid/Strong Base Titrations
A. Initial pH
This is determined by the initial concentration of the weak acid and the dissociation constant, K a.HAAOHK
a 3 Set up an ICE table for the dissociation of the weak acid in water. The "change" line will include entries like "-x" and "+x" because the weak acid will only partially dissociate. Solve for [H 3 O ] and use it to calculate the pH: pH = -log[H 3 O Be aware that polyprotic acids behave a little differently than monoprotic acids and will be discussed next week.B. pH during the titration
For every mole of OH
added to a weak acid, one mole of acid will be neutralized. Neutralization of 1 mole of a weak acid (HA) will produce one mole of water and one mole of its conjugate base (AHA + OH
H 2 O + A Since significant amounts of both the weak acid AND its conjugate base are present, the pH can be calculated using the Henderson-Hasselbalch equation: acidbasepKpH a log This can only be used after the initial point and before the equivalence point, when the acid and its conjugate base are both major species in the solution. At the half-way point, when exactly one half of the HA has been converted to A by addition of OH , pH = pK a , because log (1) = 0.C. Equivalence point
The equivalence point is reached when equal moles of acid and base are mixed. At the equivalence point, all of the HA will be converted to A , the conjugate base of the initial weak acid. The conjugate base (and water) will be the major species in the solution. There will be as many moles of these conjugate species as there were moles of acid or base at the beginning of the titration. The volume will be the sum of the initial volume of weak acid and the volume of base added to bring the solution to the equivalence point. In the case of a weak acid titrated with a strong base, A + H 2O HA + OH
The extent of this reaction will be determined by the K b of the conjugate base, A This can be calculated from the following relationship: baw KKK 14 100.1The resulting solution will be basic. The pH is calculated by setting up an ICE table for the dissociation of the conjugate base in water, and using [OH ] to solve for pOH.
D. Above the equivalence point
Above the equivalence point, any added strong base will determine the pH. The excess OH divided by the volume can be used to determine the pOH (and therefore pH) of the solution.1. Consider the titration of 50.0 mL of HNO
2 (K a = 4.0x10 -4 ) with KOH.At each step of the titration...
a) write a reaction to show the initial reaction upon mixing b) construct an ICE table to represent the reaction (Should you use concentration or moles?) c) determine the major species present after the reaction is complete d) calculate the pH of the solution Complete these steps for the addition of 0.0 mL, 25.0 mL, 50.0 mL, 90.0 mL,100.0 mL, 110.0 mL and 150.0 mL of KOH.
0.0 mL KOH
a) HNO 2 + H 2 O NO 2- + H 3 O b) [HNO 2 ] [NO 2- ] [H 3 OInitial 2.0 0 0
Change -x +x +x
Equil. 2.0 - x x x
This ICE table uses units of concentration because the volume is fixed c) The major species present at equilibrium are HNO 2 and H 2O (weak acid
solution) d) pH is calculated using the concentration of [H 3 O55.11083.2log valid)is rule (5% 1083.20.20.2100.4
22222 324
pHxx xx
HNOOHNOK
a25.0 mL KOH
a) HNO 2 + OH NO 2- + H 2 O b) This ICE table uses units of moles because the volume changes when theKOH is added
222
HNO mol 100.0L1HNO mol 2.0L 050.0HNO mol
OH mol 025.0NaOHmol1.0OH mol 1.0
L 1NaOH mol 1.0L 025.0OH mol
moles HNO 2 OH NO 2-Initial 0.100 0.025 0
Change -0.025 -0.025 +0.025
Equil. 0.075 0 0.025
c) The major species present at equilibrium are HNO 2 , NO 2- , K and H 2O (buffer
solution) d) pH is calculated using the Henderson-Hasselbalch equation40.3100.4loglog
4 aa KpK92.2075.0025.0log40.3acidbaselog
a pKpH50.0 mL KOH
a) HNO 2 + OH NO 2- + H 2 O b) This ICE table uses units of moles because the volume changes when theKOH is added
OH mol 050.0NaOHmol1.0OH mol 1.0
L 1NaOH mol 1.0L 050.0OH mol
moles HNO 2 OH NO 2-Initial 0.100 0.050 0
Change -0.050 -0.050 +0.050
Equil. 0.050 0 0.050
c) The major species present at equilibrium are HNO 2 , NO 2- , K and H 2O (buffer
solution) d) pH is calculated using the Henderson-Hasselbalch equation40.3050.0050.0log40.3acidbaselog
a pKpH **This is the halfway point, so pH = pK a90.0 mL KOH
a) HNO 2 + OH NO 2- + H 2 O b) This ICE table uses units of moles because the volume changes when theKOH is added
OH mol 090.0NaOHmol1.0OH mol 1.0
L 1NaOH mol 1.0L 090.0OH mol
moles HNO 2 OH NO 2-Initial 0.100 0.090 0
Change -0.090 -0.090 +0.090
Equil. 0.010 0 0.090
c) The major species present at equilibrium are HNO 2 , NO 2- , K and H 2O (buffer
solution) d) pH is calculated using the Henderson-Hasselbalch equation35.4010.0090.0log40.3acidbaselog
a pKpH100.0 mL KOH
a) HNO 2 + OH NO 2- + H 2 O b) This ICE table uses units of moles because the volume changes when theKOH is added
OH mol 100.0NaOHmol1.0OH mol 1.0
L 1NaOH mol 1.0L 100.0OH mol
moles HNO 2 OH NO 2-Initial 0.100 0.100 0
Change -0.100 -0.100 +0.100
Equil. 0 0 0.100
c) The major species present at equilibrium are NO 2- , K and H 2O (weak base
solution) **This is the equivalence point of the titration because the initial mole values of weak acid and strong base were equivalent d) pH is calculated using the K b for the weak base; set up a new ICE table using concentration units (the volume of this solution is fixed now) NO 2- + H 2 O OH + HNO 2 222
NO M 667.0L100.0L 050.0NO mol 0.100NO
[NO 2- ] [OH ] [HNO 2Initial 0.667 0 0
Change -x +x +x
Equil. 0.667-x x x
61.81439.51008.4log valid)is rule (5% 1008.4667.0667.0105.2100.4100.1
66222 2 11 414
pOHpHpOHxx xx
NOOHHNOK
quotesdbs_dbs20.pdfusesText_26[PDF] acid base test review answers
[PDF] acid/base stoichiometry practice problems answers
[PDF] acide acétique
[PDF] acide base ph cours
[PDF] acide base ph exercice
[PDF] acide base ph terminale s
[PDF] acide base physique chimie
[PDF] acide base physique terminale s
[PDF] acide base physique ts
[PDF] acide et base conjuguée
[PDF] acide et base de bronsted
[PDF] acide et base de lewis
[PDF] acide et base exercices corrigés pdf
[PDF] acide et base pdf
