 ME 201
ME 201
ME 201 Thermodynamics. 1. ME 201. Thermodynamics. Second Law Practice Problems. 1. Ideally which fluid can do more work: air at 600 psia and 600°F or steam at.
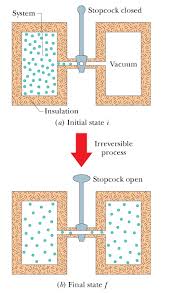 Chapter 20: Entropy and the Second Law of Thermodynamics
Chapter 20: Entropy and the Second Law of Thermodynamics
Heat does not transfer from the ice to the water (though this would not violate the law of energy conservation). AS > 0. Page 19. Sample Problem. Water is
 More problems on second law of thermodynamics
More problems on second law of thermodynamics
Understand the energy balance for a flow system and solve numericals on the applications on energy balance. Write the entropy balance equation.
 Untitled
Untitled
Thermodynamics problems: SOLUTIONS. Easy: Probability. Suppose you shuffle a dP. T=T₂. Page 15. A4 Using the differential form of the first law of ...
 1 Chapter 20 Entropy and second law of thermodynamics 1 Content
1 Chapter 20 Entropy and second law of thermodynamics 1 Content
S (extensive) F (extensive)
 Counterintuitive effect of gravity on the heat capacity of a metal
Counterintuitive effect of gravity on the heat capacity of a metal
18-Apr-2015 It is found that this solution violates the second law of thermodynamics ... problems-and-solutions/1967/1st_IPhO_1967.pdf. 15. Page 16. 2 Rudolf ...
 LECTURE NOTES ON THERMODYNAMICS
LECTURE NOTES ON THERMODYNAMICS
31-Oct-2023 The objective of the course is to survey practical and theoretical problems in classical thermodynamics. The emphasis is on the axiomatic ...
 Solutions Manual for Thermodynamics and Chemistry
Solutions Manual for Thermodynamics and Chemistry
09-Jun-2020 The sign of S is positive as predicted by the second law for an irreversible process in an isolated system. 4.5 Refer to the apparatus shown ...
 Problem Solutions
Problem Solutions
The entropy of the oxide is 51.0 kJ/K per kilomole. According to the second law of thermodynamics the entropy of a closed system suffering any transformation
 The First Law of Thermodynamics: Closed Systems Heat Transfer
The First Law of Thermodynamics: Closed Systems Heat Transfer
Solution: Assume that i) the gas is a closed system ii) the moving boundary is only But from the second law point of view
 SECOND LAW OF THERMODYNAMICS (Numerical Problem
SECOND LAW OF THERMODYNAMICS (Numerical Problem
SECOND LAW OF. THERMODYNAMICS. (Numerical Problem Solution) e-content for B.Sc Physics (Honours). B.Sc Part-I. Paper-II. Dr. Ayan Mukherjee.
 ME 201
ME 201
Thermodynamics. Second Law Practice Problems. 1. Ideally which fluid can do more work: air at 600 psia and 600°F or steam at. 600 psia and 600°F. Solution:.
 Chapter 20: Entropy and the Second Law of Thermodynamics
Chapter 20: Entropy and the Second Law of Thermodynamics
Heat does not transfer from the ice to the water (though this would not violate the law of energy conservation). AS > 0. Page 19. Sample Problem. Water is
 LECTURE NOTES ON THERMODYNAMICS
LECTURE NOTES ON THERMODYNAMICS
14-Feb-2010 These notes emphasize problem-solving and rigorous development of the ... 1850: Rudolf Julius Emanuel Clausius formalizes the second law of ...
 The First Law of Thermodynamics: Closed Systems Heat Transfer
The First Law of Thermodynamics: Closed Systems Heat Transfer
Solution: The energy content of the oven is increased during this process. a) The energy transfer to the oven is not caused by a temperature difference between
 Second Law and Entropy
Second Law and Entropy
Second law of thermodynamics - State the second law of thermodynamics as it Entropy - Solve problems involving the following concepts: (a) efficiency.
 Thermodynamic Properties and calculation
Thermodynamic Properties and calculation
First Law of Thermodynamic: ?Combining the first and second laws in reversible process ... other hand the answers to parts (a) and (b) show.
 Solution of Quiz 1: First Law of Thermodynamics
Solution of Quiz 1: First Law of Thermodynamics
12-Sept-2012 (d) The atom will undergo centre of mass motion with energy E3 ? E2. (e) The gaseous atoms collide with one another transferring the residual ...
 Problem Solutions
Problem Solutions
According to the second law of thermodynamics the entropy of a closed system suffering any transformation can not diminish.
 1 Solutions to sample quiz problems and assigned problems
1 Solutions to sample quiz problems and assigned problems
Quiz Problem 5. Use the Master equation to prove the second law of thermodynamics i.e. in a closed system. dS/dt ? 0. Solution. see lecture notes page 10.
 Second Law Problems - Michigan State University
Second Law Problems - Michigan State University
Thermodynamics Second Law Practice Problems Ideally which fluid can do more work: air at 600 psia and 600°F or steam at600 psia and 600°F Solution: The maximum work a substance can do is given by its availablity We willassume that we have a closed system so that÷
 Thermodynamics: The Three Laws of Thermodynamics - Equations
Thermodynamics: The Three Laws of Thermodynamics - Equations
The second law of thermodynamics asserts that processes occur in a certain direction and that the energy has quality as well as quantity The first law places no restriction on the direction of a process and satisfying the first law does not guarantee that the process will occur
 Chapter 7 THE SECOND LAW OF THERMODYNAMICS
Chapter 7 THE SECOND LAW OF THERMODYNAMICS
Second Law of Thermodynamics and Thermal Energy Reservoirs 7-1C Water is not a fuel; thus the claim is false 7-2C Transferring 5 kWh of heat to an electric resistance wire and producing 5 kWh of electricity 7-3C An electric resistance heater which consumes 5 kWh of electricity and supplies 6 kWh of heat to a room
 Chapter 5 The Second Law of Thermodynamics - Saylor Academy
Chapter 5 The Second Law of Thermodynamics - Saylor Academy
The second law of thermodynamics states that processes occur in a certain direction not in just any direction Physical processes in nature can proceed toward equilibrium spontaneously: Water flows down a waterfall Gases expand from a high pressure to a low pressure Heat flows from a high temperature to a low temperature
 Searches related to second law of thermodynamics problems and solutions pdf filetype:pdf
Searches related to second law of thermodynamics problems and solutions pdf filetype:pdf
2 Second law of thermodynamics If a closed system is in a configuration that is not the equilibrium configuration the most probable consequence will be that the entropy of the system will increase monotonically If an irreversible process occurs in a closed system the entropy of the system always increases; it never decreases
What are the first three laws of thermodynamics?
- There three laws are: The first law of thermodynamics is the law of the conservation of energy; it states that energy cannot be created nor destroyed. An example is when the chlorophyll absorbs light and transforms it into chemical energy.
How many laws of thermodynamics are there?
- Traditionally, thermodynamics has recognized three fundamental laws, simply named by an ordinal identification, the first law, the second law, and the third law. [1] [2] [3] A more fundamental statement was later labelled as the zeroth law, after the first three laws had been established.
What does first law of thermodynamics mean?
- The first law of thermodynamics, also known as the law of conservation of energy states that energy can neither be created nor destroyed, but it can be changed from one form to another. According to this law, some heat given to the system is used to change the internal energy while the rest is used in doing work by the system.
[PDF] second order differential equation rlc circuit
[PDF] second order low pass filter
[PDF] second trimester bleeding differential diagnosis
[PDF] second year computer engineering syllabus pune university 2019 20
[PDF] secondary amine reaction with hcl
[PDF] secondary amine reaction with nano2 and hcl
[PDF] secondary amine reaction with water
[PDF] secondary colors of light
[PDF] secondary consumers are eaten by larger
[PDF] secondary consumers in the desert
[PDF] secondary consumers in the ocean
[PDF] secondary consumers in the rainforest
[PDF] secondary consumers in the savanna
[PDF] secondary consumers in the tundra
