 Solutions
Solutions
Chemistry the partial vapour pressure of each component of the solution is Another important class of solutions consists of solids dissolved in liquid ...
 D:TEXTBOOKSRATIONALISED 20222-23NehaP85
D:TEXTBOOKSRATIONALISED 20222-23NehaP85
V. 0.130. = 3.17 V – 0.21V = 2.96 V. Solution. Rationalised 2023-24. Page 10. 40. Chemistry.
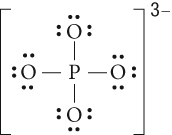 Section 4.1: Types of Chemical Bonds
Section 4.1: Types of Chemical Bonds
Page 12. Copyright © 2012 Nelson Education Ltd. Chapter 4: Chemical Bonding 7. Answers will vary. Sample answer: Coordinate covalent bonding is like you ...
 Chem 12 SM Ch5 Review final new ok revised
Chem 12 SM Ch5 Review final new ok revised
Δ = −. Statement: The enthalpy change ΔH
 Chem12 SM Ch05 Section5 3 final ok revised
Chem12 SM Ch05 Section5 3 final ok revised
Section 5.3 Questions page 313. 1. (a) Solution: Step 1: Use the balanced chemical equation to determine the bonding of each substance. H2(g) +
 Chem12 SM Ch05 Section5 2 final ok revised
Chem12 SM Ch05 Section5 2 final ok revised
Tutorial 3 Practice page 304. 1. (a) Solution: Step 1: Write the balanced chemical equation without the energy term. 2 C2H2(g) + 5 O2
 Grade 12 Chemistry: A Foundation for Implementation
Grade 12 Chemistry: A Foundation for Implementation
Jan 21 2011 ... textbook. The successful implementation of Grade 12 Chemistry depends ... solutions based on this result? Explain. 3. Are there any reactions ...
 Chem12 SM Ch7 Section7.5 final ok revised
Chem12 SM Ch7 Section7.5 final ok revised
Solution: Step 1. Convert the given initial amount of cyclopropane gas to (c) The chemical system is not at equilibrium. We have been given K = 1.2 × 102 ...
 Section 8.4: Calculations Involving Acidic Solutions
Section 8.4: Calculations Involving Acidic Solutions
Section 8.4: Calculations Involving Acidic Solutions. Tutorial 1 Practice page x2 ≈ 6.2 × 10−12 x = [H+(aq)]. ≈ 2.49 × 10−6 mol/L. pH = −log(2.49 × 10 ...
 Chem12 SM Ch8 Section8e1 final ok revised
Chem12 SM Ch8 Section8e1 final ok revised
solution; an Arrhenius base forms hydroxide ions in aqueous solution. (b) A Brønsted–Lowry acid is a proton donor; a Brønsted–Lowry base is a proton.
 Grade 12 Chemistry: A Foundation for Implementation
Grade 12 Chemistry: A Foundation for Implementation
Jan 21 2011 promotes the idea that all answers are enshrined in a textbook. The successful implementation of Grade 12 Chemistry depends on a ...
 Chem12 SM Ch7 Section7.6 final revised
Chem12 SM Ch7 Section7.6 final revised
Chapter 7: Chemical Equilibrium 7.6-1 Write a balanced equation for the solution equilibrium. Ca3(PO4)2(s) ! "!# !! ... Ksp of CuI(s) = 1.3 × 10?12.
 Chem12 SM Ch05 Section5 2 final ok revised
Chem12 SM Ch05 Section5 2 final ok revised
Chapter 5: Thermochemistry. 5.2-3. Solution: Step 1: Determine the change in temperature AT . final initial. 27.8 °C 19.8 °C.
 Chem 12 SM Ch5 Review final new ok revised
Chem 12 SM Ch5 Review final new ok revised
(a) Answers may vary. Sample answer: Hydrogen gas has a high enthalpy of combustion releasing about 2.5 times the quantity of energy per gram than methane but
 Solutions
Solutions
Chemistry. Type of Solution. Solute. Solvent. Common Examples. Gaseous Solutions. Gas. Gas. Mixture of oxygen and nitrogen gases.
 Chem12 SM Ch7 Section7.5 final ok revised
Chem12 SM Ch7 Section7.5 final ok revised
Chapter 7: Chemical Equilibrium Solution: N2O4(g) ! "!# !! 2 NO2(g) ... Using the balanced chemical equation construct an ICE table for calculating.
 Chem12 SM Ch05 Section5.4 final ok revised
Chem12 SM Ch05 Section5.4 final ok revised
An enthalpy diagram of the reaction is: Page 2. Copyright © 2012 Nelson Education Ltd. Chapter 5: Thermochemistry. 5.4-2. 2. (a) Solution: Step 1: Label the
 chemistry grade 12 studen textbook
chemistry grade 12 studen textbook
CHEMISTRY GRADE 12. 2. MAIN CONTENTS. 1.1 Homogeneous and Heterogeneous Mixtures. 1.2 Types of Solutions. 1.3 The Solution Process.
 EngLinks
EngLinks
Grade 12 Chemistry Review Workbook. 1 of 22. 1. Organic Chemistry. General IUPAC rules for naming organic compounds: 1) The lowest numbers possible must be
 Chem12 SM Ch05 Section5 3 final ok revised
Chem12 SM Ch05 Section5 3 final ok revised
combustion of ethene gas to gaseous carbon dioxide and liquid water is an exothermic reaction. 3. Solution: Step 1: Use the balanced chemical equation:.

Copyright © 2012 Nelson Education Ltd. Chapter 7: Chemical Equilibrium 7.6-1 Section 7.6: Solubility Equilibria and the Solubility Product Constant Tutorial 1 Practice, page 462 1. Solution: [MgCO3(s)] is constant, so it is not included in the equation. Therefore, Ksp = [Mg2+(aq)][CO32-(aq)] Statement: The solubility product constant equation for the solubility equilibrium is Ksp = [Mg2+(aq)][CO32-(aq)] 2. Given: iron(III) hydroxide dissolves in water Required: Ksp equation Solution: Step 1. Write a balanced equation for the dissolution of iron(III) hydroxide in water. Fe(OH)3(s)
Fe+(aq) + 3 OH-(aq) Step 2. [Fe(OH)3(s)] is constant, so it is not included in the equation. Therefore, Ksp = [Fe+(aq)][OH-(aq)]3 Statement: The solubility product constant equation for iron(III) hydroxide is Ksp = [Fe+(aq)][OH-(aq)]3 3. Given: At 25 ºC: [Ca2+(aq)] = 4.53 × 10-7 mol/L; [PO43-(aq)] = 3.02 × 10-7 mol/L Required: Ksp of calcium phosphate at 25 ºC Solution: Step 1. Write a balanced equation for the solution equilibrium. Ca3(PO4)2(s)
3 Ca2+(aq) + 2 PO43-(aq) Step 2. Write the solubility constant equation for the equilibrium, omitting any solids. Ksp = [Ca2+(aq)]3[PO43-(aq)]2 Step 3. Substitute the given ion concentrations into the Ksp equation and solve. K
sp =[Ca 2+ (aq)] 3 [PO 4 !!3! (aq)] 2 =(4.53!"!10 !7 3 (3.02!"!10 !7 2 K sp =8.48!"!10 !33 Statement: The Ksp of calcium phosphate in a saturated solution at 25 °C is8.48 ! 10
"33Tutorial 2 Practice, page 464 1. Given: solubility of MgF2(s) = 1.72 × 10-3 g/100 mL at 25 ºC Required: Ksp of MgF2(s) at 25 ºC Solution: Step 1. Write the balanced equation. MgF2(s)
Mg2+(aq) + 2 F-(aq) Step 2. Write the solubility product constant equation. K sp =[Mg 2+ (aq)][F (aq)] 2Step 3. Determine the equilibrium concentrations in mol/L of the dissolved ions. Use an ICE table. Since the concentration of solid MgF2 remains constant, it is not included.
Copyright © 2012 Nelson Education Ltd. Chapter 7: Chemical Equilibrium 7.6-2 MgF2(s)Mg2+(aq) + 2 F-(aq) I - 0.00 0.00 C - +x +2x E - x 2x Step 4. Calculate all concentrations in mol/L. To convert given mass solubility of solid magnesium fluoride to molar solubility, first calculate the molar mass of magnesium fluoride. Then, use the molar mass to convert mass solubility to molar solubility. molar mass of MgF
2 =24.31! g mol +(2)19.00! g mol molar mass of MgF 2 =62.31 g/mol molar solubility of MgF 21.72 ! 10
"3 g100 mL
1 mol62.31 g
1000 mL
1 L molar solubility of MgF 2 =2.76!10 "4 mol/LStep 5. Write the Ksp equation using the expressions in the ICE table, and then substitute the molar solubility of solid magnesium fluoride for x. K
sp =[Mg 2+ (aq)][F (aq)] 2 =(x)(2x) 2 =(2.76!!!10 "4 )(5.52!!!10 "4 2 K sp =8.41!!!10 "11 Statement: The solubility product constant of solid magnesium fluoride is8.41 ! 10
"112. Given: Ksp of AgI(s) at 25 ºC = 8.5 × 10-17 Required: molar solubility of AgI(s) at 25 ºC Solution: Step 1. Write the balanced chemical equation. AgI(s)
Ag+(aq) + I-(aq) Step 2. Write the solubility product constant equation. K sp =[Ag (aq)][I (aq)] [Ag (aq)]=[I (aq)] K sp =[Ag (aq)] 2Step 3. Determine the equilibrium concentrations of the dissolved ions. From the balanced equation, the molar ratio is 1:1:1, so the concentrations of these ions are equal at any point in time. Since the solubility of solid silver iodide is 8.5 × 10-17 mol/L, then a maximum of 8.5 × 10-17 mol of solid silver iodide can dissolve in water to produce 1.0 L of solution at 25 °C. This will form 8.5 × 10-17 mol of silver ions and 8.5 × 10-17 mol of iodide ions. Therefore, the equilibrium concentrations of these ions are also equal to 8.5 × 10-17 mol/L. Step 4. Substitute the equilibrium concentrations into the Ksp equation and solve for Ksp.
Copyright © 2012 Nelson Education Ltd. Chapter 7: Chemical Equilibrium 7.6-3 K sp =[Ag (aq)] 28.5!!!10
"7 =[Ag (aq)] 2 [Ag (aq)]=8.5!!!10 "17 [Agquotesdbs_dbs2.pdfusesText_2[PDF] chemistry notes for class 12 pdf
[PDF] chiffres coronavirus france 11 mai
[PDF] chiffres coronavirus france 11 mai 2020
[PDF] chiffres covid france 6 juin
[PDF] child care cost per province
[PDF] child language acquisition stages
[PDF] childhood in france vs us
[PDF] china paris agreement goals
[PDF] china population 2019 vs us
[PDF] chinese food near me open
[PDF] chinese language cantonese vs mandarin
[PDF] chinese restaurant in paris tx
[PDF] choix heure ete ou hiver france
[PDF] chômage technique code travail maroc
