 Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
Aldehydes Aldehydes Ketones and Carboxylic Carboxylic Acids
most important functional groups in organic chemistry. In aldehydes the carbonyl group is bonded They add fragrance and flavour to nature
 The Carbonyl Group Nomenclature of Aldehydes and Ketones
The Carbonyl Group Nomenclature of Aldehydes and Ketones
the parent compound benzaldehyde. (The carbon to which the aldehyde group is attached is carbon “1”). 10. Examples: Naming Aldehydes and Ketones.
 INFRARED SPECTROSCOPY (IR)
INFRARED SPECTROSCOPY (IR)
Uses of the Infrared Spectrum (p. 847-853). • Look over pages 853-866 after viewing this presentation for additional examples of various functional groups.
 New Aldehyde?Functional Methacrylic Water?Soluble Polymers
New Aldehyde?Functional Methacrylic Water?Soluble Polymers
there are no literature examples of water-soluble aldehyde- functional vinyl monomers. Aldehydes are extremely useful functional groups in.
 Infrared Spectroscopy
Infrared Spectroscopy
15 mai 2013 The exact wavenumber of the C=O stretch can give you clues as to whether the compound is a ketone aldehyde
 Short Summary of IUPAC Nomenclature of Organic Compounds
Short Summary of IUPAC Nomenclature of Organic Compounds
Nomenclature of Molecules Containing Substituents and Functional Groups hydroxy- amino-. Structure. Family of Compound. Carboxylic Acid. Aldehyde.
 Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
Aldehydes Aldehydes Ketones and Carboxylic Acids Aldehydes
most important functional groups in organic chemistry. In aldehydes the carbonyl group is bonded They add fragrance and flavour to nature
 Subject: Chemistry Synthesis Key features & characteristics
Subject: Chemistry Synthesis Key features & characteristics
2 Example of making an aldehyde. Oxidation of primary alcohol to aldehyde group. Isomerism. Aldehydes and ketones are functional group.
 1.1 Functional Groups of Biomolecules and their Reactions
1.1 Functional Groups of Biomolecules and their Reactions
1) which contributes to the reactivity of the compounds that have this functional group (aldehydes
 New Aldehyde?Functional Methacrylic Water?Soluble Polymers
New Aldehyde?Functional Methacrylic Water?Soluble Polymers
there are no literature examples of water-soluble aldehyde- functional vinyl monomers. Aldehydes are extremely useful functional groups in.
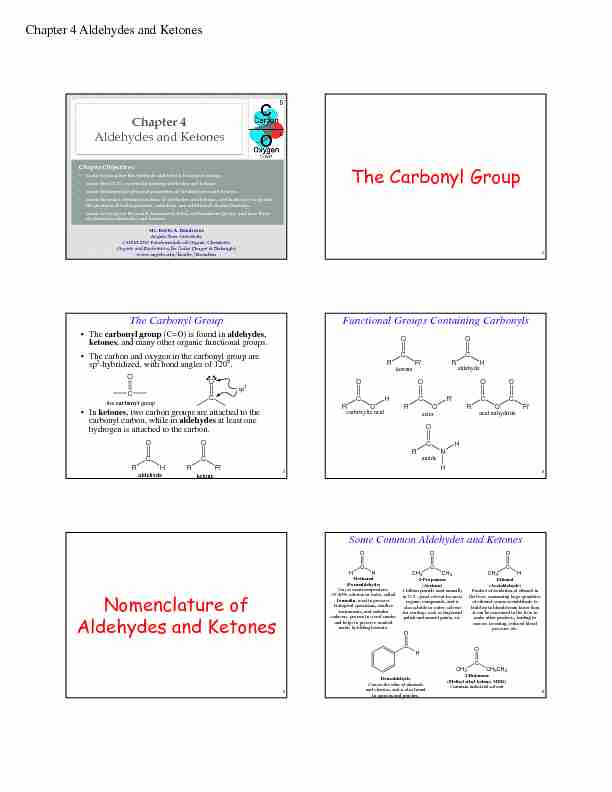
Chapter 4 Aldehydes and Ketones
Mr. Kevin A. Boudreaux
Angelo State University
CHEM 2353 Fundamentals of Organic Chemistry
Organic and Biochemistry for Today (Seager & Slabaugh) www.angelo.edu/faculty/kboudreaChapter Objectives:
•Learn to recognize the aldehyde and ketone functional groups. •Learn the IUPAC system for naming aldehydes and ketones. •Learn the important physical properties of the aldehydes and ketones. •Learn the major chemical reaction of aldehydes and ketones, and learn how to predict the products of hydrogenation, oxidation, and addition of alcohol reactions. •Learn to recognize the acetal, hemiacetal, ketal, and hemiketal group, and how these are related to aldehydes and ketones.Chapter 4Aldehydes and Ketones
The Carbonyl Group
2The Carbonyl Group
• The carbonyl group (C=O) is found in aldehydes, ketones, and many other organic functional groups.
• The carbon and oxygen in the carbonyl group are sp2 -hybridized, with bond angles of 120°.•In ketones, two carbon groups are attached to the carbonyl carbon, while in aldehydesat least one hydrogen is attached to the carbon.CO
sp 2 CO the carbonyl group C RR'O C RHO ketonealdehyde 3Functional Groups Containing CarbonylsC
RR'O C RHO C ROO HC ROO R' C ROO C R'O C RNO H H ketonealdehyde carboxylic acid esteracid anhydride amide 4Nomenclature of
Aldehydes and Ketones
5Some Common Aldehydes and KetonesHC
HOMethanal
(Formaldehyde)Gas at room temperature;
35-40% solution in water, called
formalin, used to preserve biological specimens, sterilize instruments, and embalm cadavers; present in wood smoke, and helps to preserve smoked meats by killing bacteria. CH 3 C CH 3 O2-Propanone
(Acetone)1 billion pounds used annually
in U.S.; good solvent for most organic compounds, and is also soluble in water; solvent for coatings such as fingernail polish and enamel paints, etc.CH 3 C HOEthanal
(Acetaldehyde)Product of oxidation of ethanol in
the liver; consuming large quantities of ethanol causes acetaldehyde to build up in bloodstream faster than it can be consumed in the liver to make other products, leading to nausea, sweating, reduced blood pressure, etc.C HOBenzaldehyde
Causes the odor of almonds
and cherries, and is also found in apricots and peaches. CH 3 C CH 2 CH 3 O2-Butanone
(Methyl ethyl ketone, MEK) Co mmon industrial solvent 6Chapter 4 Aldehydes and Ketones
Nomenclature of Aldehydes
• Select the longest carbon chain containing the carbonyl carbon. • The -eending of the parent alkane name is replaced by the suffix -al.quotesdbs_dbs2.pdfusesText_2[PDF] aldehyde functional group ir
[PDF] aldehyde functional group ir spectrum
[PDF] aldehyde functional group name
[PDF] aldehyde functional group properties
[PDF] aldehyde functional group suffix
[PDF] aldehyde hydrolysis
[PDF] aldehyde ir spectrum
[PDF] aldehyde ketone and carboxylic acid mcq pdf
[PDF] aldehyde ketone and carboxylic acid notes for neet pdf
[PDF] aldehyde ketone and carboxylic acid notes in hindi
[PDF] aldehyde ketone and carboxylic acid notes pdf download
[PDF] aldehyde ketone and carboxylic acid pdf target
[PDF] aldehyde ketone and carboxylic acid questions pdf
[PDF] aldehyde to ketone
