 INORGANIC C B. Sc. I YEAR INORGANIC CHEMISTRY CHEMISTRY-I
INORGANIC C B. Sc. I YEAR INORGANIC CHEMISTRY CHEMISTRY-I
Unit Written By. Unit No. 1. Dr. K. S. Dhami (Ret. Proff.) 01 02
 Modern inorganic chemistry
Modern inorganic chemistry
Page 1. Modern inorganic chemistry. AN INTERMEDIATE TEXT. C. CHAMBERS B.Sc in first-year tertiary level chemistry courses. The new syllabuses have made it ...
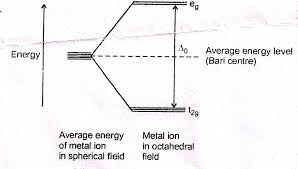 B. Sc. II YEAR INORGANIC CHEMISTRY-II
B. Sc. II YEAR INORGANIC CHEMISTRY-II
INORGANIC CHEMISTRY-II. BSCCH-201. UTTARAKHAND OPEN UNIVERSITY. Page 1. UNIT 1: CHEMISTRY OF THE ELEMENTS OF. FIRST TRANSITION (3-d) SERIES. CONTENTS: 1.1
 STATE MODEL SYLLABUS FOR UNDER GRADUATE COURSE IN
STATE MODEL SYLLABUS FOR UNDER GRADUATE COURSE IN
Chand Publisher. 2012. Reference Books: 1. Y R Sharma
 1 INDIRA GANDHI UNIVERSITY MEERPUR (REWARI)
1 INDIRA GANDHI UNIVERSITY MEERPUR (REWARI)
B. Sc IIIrd Semester. Paper IX (Theory) Inorganic Chemistry. Max. Marks: 29. CH-301. Time: 3 Hrs
 INORGANIC CHEMISTRY
INORGANIC CHEMISTRY
B.Sc. B.Sc. (Hons.) & M.Sc. Students of Indian Universities as also for Aqueous chemistry of copper (1) (680). Aqueous chemistry of Cu (II) (681).
 Kurukshetra University Kurukshetra Scheme and Syllabi for B.Sc
Kurukshetra University Kurukshetra Scheme and Syllabi for B.Sc
II Year (IIIrd Semester). Paper-VIII (CH-201) Inorganic Chemistry (Theory). M.Marks: 32. Time: 3 Hrs. Note: Nine questions will be set. Q.No.1 based on whole
 Concise Inorganic Chemistry (4th Edition)
Concise Inorganic Chemistry (4th Edition)
Aparl from ant fair dealing' for the purposes of resean:h 1.>r private study or criticism or review. as permitted under the UK Copyright Designs and. Patents
 B.Sc. III YEAR INORGANIC CHEMISTRY-III
B.Sc. III YEAR INORGANIC CHEMISTRY-III
As a mixture of biochemistry and inorganic chemistry bioinorganic chemistry is 8.3.1 Classification of inorganic polymers. 8.3.1 General properties of ...
 INORGANIC C B. Sc. I YEAR INORGANIC CHEMISTRY CHEMISTRY-I
INORGANIC C B. Sc. I YEAR INORGANIC CHEMISTRY CHEMISTRY-I
INORGANIC CHEMISTRY-I. BSCCH-101. UTTARAKHAND OPEN UNIVERSITY. Page 1 (v) By the year 1900 some 30 more elements had been added to the list of elements ...
 Modern inorganic chemistry
Modern inorganic chemistry
in first-year tertiary level chemistry courses. the facts of inorganic chemistry and in this book the first four chap- ... (Liverpool B.Sc.
 B. SC. CHEMISTRY (Subsidiary)
B. SC. CHEMISTRY (Subsidiary)
B.Sc. COURSE PLAN (Subsidiary). Inorganic Chemistry. 1st Year. 1st Semester : Paper IS & Paper IIS (Group C each). Marks : 16 + 16.
 Notes of inorganic chemistry for BSc II
Notes of inorganic chemistry for BSc II
(1). T on the basis of this concept have been defined in torms of aqueous solutions. This defination is applicable for hydrogen and hydroxyl Compounds.
 B. Sc. II YEAR INORGANIC CHEMISTRY-II
B. Sc. II YEAR INORGANIC CHEMISTRY-II
Unit-1 Chemistry of elements of first transition series INORGANIC CHEMISTRY-II. BSCCH-201. UTTARAKHAND OPEN UNIVERSITY. Page 1 ... ion_compounds.pdf.
 Syllabus B.Sc.-Chemistry
Syllabus B.Sc.-Chemistry
[1]. V. B. S. Purvanchal University Jaunpur. Syllabus. B.Sc.-Chemistry. B.Sc.-1- 1. Inorganic Chemistry. Theoretical. 50. 3.00. 2. Organic Chemistry.
 MODEL PAPER FIRST YEAR B.Sc. DEGREE EXAMINATION
MODEL PAPER FIRST YEAR B.Sc. DEGREE EXAMINATION
MODEL PAPER. FIRST YEAR B.Sc. DEGREE EXAMINATION. SEMESTER-I. CHEMISTRY Course-I: INORGANIC & PHYSICAL CHEMISTRY. Time: 3 hours. Maximum Marks: 75.
 t- * rr1*
$---
t- * rr1*
$---
B. Sc. Ist Year (Ist Semester) paper I (Theory) Inorganic chemistry (cH-101) Max. Marks: 27. Time: 3 Hrs. N o t e: The examiners will set seven questions in
 Inorganic Chemistry (paper code - CH-101) B.Sc 1st Year (Sem. 1st)
Inorganic Chemistry (paper code - CH-101) B.Sc 1st Year (Sem. 1st)
Inorganic Chemistry (paper code - CH-101). B.Sc 1st Year (Sem. 1st). GDC Memorial College. (Approved by Govt. of Haryana & Affiliated to M D University
Unit-I
Atomic Structure
Idea of de Broglie matter waves, Heisenberg uncertainty principle, atomic orbitals, quantum numbers, radial and angular wave functions and probability distribution curves, shapes of s, p, d orbitals.Unit-II
Periodic Properties
General principles of periodic table: Aufbau and Pauli exclusion principles, Hund's multiplicity rule. Electronic configurations of the elements, effectivenegativity definition, methods of determination or evaluation, trends in periodic table (in s &p block
elements).Unit-III
Covalent Bond
Valence bond theory and its limitations, directional characteristics of covalent bond, various types of
hybridization and shapes of simple inorganic molecules and ions ( BeF2, BF3, CH4, PF5, SF6,IF7 SO42 -, ClO4- )Valence shell electron pair repulsion (VSEPR)5 theory to NH3, H3O+, SF4, CIF3, ICI2- and H2O. MO theoryof heteronuclear (CO and NO) diatomic.molecules, , bond strength and bond energy, pe rcentage ionic character from dipole moment and electronegativity difference.Unit-IV
Ionic Solids
Ionic structures (NaCl,CsCl, ZnS(Zinc Blende), CaF2) radius ratio effect and coordination number, limitation of radius ratio rule, lattice defects, semiconductors, lattice energy (methamtical derivation exc luded) and Born-Haber cycle, solvation ene rgy and its relation with solubility of ionic solids, polarizing power and polarisability of ions, Fajan's rule. Department of Chemistry Inorganic Chemistry (paper code - CH-101)B.Sc 1st Year (Sem. 1st) GDC Memorial College
(Approved by Govt. of Haryana & Affiliated to M D University, Rohtak)Bahal - 127 028 (Distt. Bhiwani) Haryana
Unit-I
Gaseous States
mean square velocity, average velocity and most probable velocity. Collision diameter, co llision number, col lision frequency and mean free path. Deviation of Real gases fromUnit-II
Critical Phenomenon: Critical temperature, Critical pressure, critical volume and their determination. PV isotherms of real gases, continuity of states, the isotherms of Vander Critical compressibility fac tor. The Law of corresponding states. Lequifaction of gases.Unit-III
Liquid States
Structure of liquids. Properties of liquids surface tension, viscosity vapour pressure and optical rotations and their determination.Unit-IV
Solid State
Classification of solids, Laws of crystallography
(i) Law of constancy of interfacial angles (ii) Law of rationality of indices (iii) Law of symmetry. Symmetry elements of crysta ls. Definition of unit cell & space lattice. Bravais lattices, crystal system. Xray diffraction by crystals. Derivation of Bragg equation. Deter- mination o f crystal structure o f NaCl, KCl. Liquid crystals: Difference between solids, liquids and liquid crystals,types of liquid crystals. Applications of liquid crystals. Department of Chemistry Physical Chemistry (paper code - CH-102)B.Sc 1st Year (Sem. 1st) GDC Memorial College
(Approved by Govt. of Haryana & Affiliated to M D University, Rohtak)Bahal - 127 028 (Distt. Bhiwani) Haryana
Unit-I
1. Structure and Bonding
Localized and delocalized chemical bond, van der Waals in teractions, resonance: conditions, resonance effect
and its applications, hyperconjugation, inductive effect, Electromeric effect & their comparison.2. Stereochemistry of Organic Compounds-I
Concept of isomerism. Types of isomerism. Optical isomerism, elements of symmetry, molecular chirality, enan-
tiomers, stereogenic centre, optical activity, properties of enantiomers, chiral and achiral molecules with two
stereogenic centres, diastereomers, threo and eryth ro diastereomers, meso compounds, resolution of enanti-
omers, inversion, retention and racemization.Unit-II
Stereochemistry of Organic Compounds-II
Relative and absolute configuration, sequence rules, R & S systems of nomenclature. Geometric isomerism de-
termination of configuration of geometric isomers. E & Z system of nomenclature, Conformational isomerism
conformational analysis of ethane and n-butane, conformations of cyclohexane, axial and equatorial bonds,.
Newman projection and Sawhorse formulae, Difference between configuration and conformation.Unit-III
quotesdbs_dbs7.pdfusesText_5[PDF] inscription administrative paris 7 paces
[PDF] inscription cnc 2020
[PDF] inscription concours ecricome 2020
[PDF] inscription dakar
[PDF] inscription faculté de médecine paris 7
[PDF] inscription licence psychologie ied paris 8
[PDF] inscription maternelle 4 ans laval
[PDF] insead
[PDF] insert into table sql
[PDF] insert numeric values sql
[PDF] inside listening and speaking pdf
[PDF] inside tokyo itinerary
[PDF] install fontspec package latex
[PDF] install fortigate

