 URGENT MEDICAL DEVICE RECALL
URGENT MEDICAL DEVICE RECALL
16 апр. 2020 г. BD is conducting a voluntary medical device recall for multiple lots of the BD PosiFlush™ SF (Sterile ... Please assist BD with assuring these ...
 Briefvorlage BD GmbH mit Logo
Briefvorlage BD GmbH mit Logo
20 апр. 2018 г. BD PosiFlush™ Heparin Lock Flush Syringes. 306509 306510
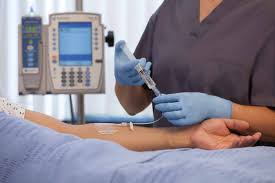 BD_Posi-Flush Brochure Option -V8 (I) (with updated CTA no)
BD_Posi-Flush Brochure Option -V8 (I) (with updated CTA no)
to help reduce complications and improve patient care. BD VAM assessments establish the baseline 5 mL BD PosiFlush™ Heparin Lock Flush Syringe 50 usp units/5 ...
 BD PosiFlushTM Pre-Filled Flush Syringes
BD PosiFlushTM Pre-Filled Flush Syringes
BD PosiFlush™ helps reduce the risk of catheter related bloodstream BD Medical. The Danby Building. Edmund Halley Road. Oxford Science Park. OXFORD OX4 4DQ.
 BD PosiFlush™
BD PosiFlush™
BD Luer-Lok™ Tip Cap: Tighter seal for closure integrity helping prevent touch contamination. Clear labelling helps reduce the risk of medication errors.
 BD PosiFlush™ Prefilled Saline Syringe Supply Update
BD PosiFlush™ Prefilled Saline Syringe Supply Update
Dear BD Valued Customer. We would like to provide you with confidence that BD is working diligently to support your prefilled flush syringe needs through our
 BD Medical Medical Surgical Systems Catalogue Canadian Version
BD Medical Medical Surgical Systems Catalogue Canadian Version
BD Activation-Assist™ technology for fast and easy needle tip shielding more than twice as many BD PosiFlush 3 mL size syringes into a 5.1 L In-Room ...
 BD Medical – Recall of heparin lock flush and normal saline syringes
BD Medical – Recall of heparin lock flush and normal saline syringes
20 апр. 2018 г. The BD PosiFlush heparin lock flush syringe is intended to help maintain patency by locking vascular access devices. • The BD pre-filled ...
 Think BD PosiFlush™ Syringes filled with Saline.
Think BD PosiFlush™ Syringes filled with Saline.
BD PosiFlush™ SP Syringes are not intended for use on a sterile field. Page 3. Silicone Lubricant. Medical-grade silicone oil is applied to the stopper and
 TABLE OF CONTENTS
TABLE OF CONTENTS
2 февр. 2017 г. The 0.9% Sodium Chloride Injection USP
 Briefvorlage BD GmbH mit Logo
Briefvorlage BD GmbH mit Logo
20 Apr 2018 bd.com. URGENT MEDICAL DEVICE RECALL. BD POSIFLUSH™ HEPARIN LOCK FLUSH ... in Attachment B to aid you with the identification of the.
 BD PosiFlush Recall
BD PosiFlush Recall
16 Apr 2020 BD is conducting a voluntary medical device recall for multiple lots of the BD PosiFlush™ SF. (Sterile Field) Saline Flush Syringe 10mL ...
 Briefvorlage BD GmbH mit Logo
Briefvorlage BD GmbH mit Logo
bd.com. URGENT MEDICAL DEVICE RECALL. BD POSIFLUSH™ PRE-FILLED NORMAL SALINE SYRINGE If you require further assistance please contact: Contact.
 URGENT MEDICAL DEVICE RECALL
URGENT MEDICAL DEVICE RECALL
16 Apr 2020 BD is conducting a voluntary medical device recall for multiple lots of the BD PosiFlush™ SF. (Sterile Field) Saline Flush Syringe 10mL ...
 Briefvorlage BD GmbH mit Logo
Briefvorlage BD GmbH mit Logo
20 Apr 2018 bd.com. URGENT MEDICAL DEVICE RECALL. BD POSIFLUSH™ HEPARIN LOCK FLUSH ... in Attachment B to aid you with the identification of the.
 BD PosiFlushTM - Because so much is on the line
BD PosiFlushTM - Because so much is on the line
The BD PosiFlush™ Prefilled Saline Syringe is a solution that is part of the BD Organisation for Standardisation (ISO) 13485 standard on Medical.
 Think BD PosiFlush™ Syringes filled with Saline.
Think BD PosiFlush™ Syringes filled with Saline.
BD PosiFlush™ SP Syringes filled with Saline
 BD Medical – Recall of heparin lock flush and normal saline syringes
BD Medical – Recall of heparin lock flush and normal saline syringes
20 Apr 2018 The BD PosiFlush heparin lock flush syringe is intended to help maintain patency by locking vascular access devices. • The BD pre-filled normal ...
 AUG 2 6 2004 000016
AUG 2 6 2004 000016
6 Feb 2004 USP BD Posiflush SF Pre-Filled Flush Syringe was modified by changing the current ... 1976 the enactment date of the Medical Device.
 BD_Posi-Flush Brochure Option -V8 (I) (with updated CTA no)
BD_Posi-Flush Brochure Option -V8 (I) (with updated CTA no)
to provide clinical benefits to help improve patient care manufacture over 14 billion BD PosiFlush™ Sterile Path Syringes. Saline and syringes.
MDS-19-1701 Page 1 of 3
BD2100 Derry Road West, Unit 100
Mississauga, Ontario L5N 0B3
bd.comURGENT MEDICAL DEVICE RECALL
BD POSIFLUSH™ PRE-
FILLED NORMAL SALINE SYRINGE
October 2019
For the Attention of:
Customer
Description of the problem and health hazard(s):
BD is initiating a voluntary recall for the BD Posiflush™ Pre-Filled Normal Saline Syringes catalog
and lot numbers referenced in the table above. The lots are being recalled as BD has become aware that a limited number of syringes labeled "Posiflush Experimental Product" and "Not for Human Use" being mixed with standard BD Posiflush™ Pre-Filled Normal Saline Syringes. BD has not received any reports of related adverse events and has confirmed that these syringes were manufactured and sterilized in accordance with our normal manufacturing process, perform as intended and do not pose any risk to health. BD is voluntarily recalling these devices to avoid confusion on the part of the user.Distribution of the affected lot
s was between June - September, 2019. Our records indicate you may have received the affected product.Please Take the Following Actions:
1. Immediately review your inventory for the specific Catalogue (Ref) and lot numbers listed
above. Destroy all product subject to the recall following your institution's process for destruction.2. Share this recall notification with all users of the product within your facility to ensure that
they are also aware of this recall.3. Complete the attached Customer Response Form and return to the contact noted on the
form whether or not you have any of the impacted material so that your receipt of this notification may be acknowledgedActions Taken by BD:
1. Corrective actions have been initiated to prevent recurrence of the identified root cause.
2. Your distributor will issue credit to you for product destroyed, once they receive the
response form.Contact Information:
If you require further assistance, please contact: Contact Contact Information Customer/Technical support (866) 979-9408Product Name Cat. No. (Ref) Lot
Nos.Exp. Date BD Posiflush™ Pre-Filled Normal
Saline Syringes 306547 9142881 30-April-2022
306575 9175971 30-June-2022
MDS-19-1701 Page 2 of 3
BD2100 Derry Road West, Unit 100
Mississauga, Ontario L5N 0B3
bd.com BD is committed to advancing the world of health. Our primary objectives are patient and user safety and providing you with quality products. We apologize for any inconvenience this issue may have caused you and thank you in advance for helping us to resolve this matter as quickly and effectively as possible.Sincerely,
For Blair Johnston Patricia Norman Vice President Director Medication Delivery Solutions (MDS) Regulatory Affairs & Quality Management BECTON DICKINSON CANADA INC. BECTON DICKINSON CANADA INC.quotesdbs_dbs26.pdfusesText_32[PDF] BD Tryptic Soy Broth (TSB)
[PDF] BD-FIL 2014 - Horaires des dédicaces du vendredi 12 septembre - Festival
[PDF] BD-jeunesse_2015-04
[PDF] BD-S1065_FICHE:Mise en page 1
[PDF] Bd. 72 1972 Quellen und Forschungen aus italienischen
[PDF] BDAF120_Decembre2003
[PDF] BDC - Anciens Et Réunions
[PDF] BDC 2014 - Amitié Solidarité Savoie - France
[PDF] bdc 2015 - CE Legrand Lagord - Anciens Et Réunions
[PDF] bdc AGRO - Conception
[PDF] BDC Beaux-Livres réassort - Anciens Et Réunions
[PDF] BDC CIVILITE 2014
[PDF] BDC FDE - Anciens Et Réunions
[PDF] bdc flammes pub - Logiciels Graphiques
